Calculate the heat content in a two tonne (2,000 kg) iron magnet at 300 K. How...
Fantastic news! We've Found the answer you've been seeking!
Question:
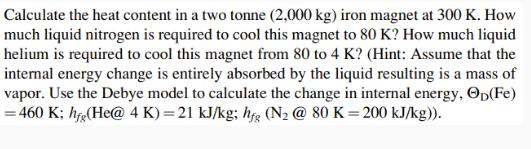
Transcribed Image Text:
Calculate the heat content in a two tonne (2,000 kg) iron magnet at 300 K. How much liquid nitrogen is required to cool this magnet to 80 K? How much liquid helium is required to cool this magnet from 80 to 4 K? (Hint: Assume that the internal energy change is entirely absorbed by the liquid resulting is a mass of vapor. Use the Debye model to calculate the change in internal energy, (Fe) = 460 K; hfg(He@ 4 K)=21 kJ/kg; hfg (N₂ @ 80 K=200 kJ/kg)). Calculate the heat content in a two tonne (2,000 kg) iron magnet at 300 K. How much liquid nitrogen is required to cool this magnet to 80 K? How much liquid helium is required to cool this magnet from 80 to 4 K? (Hint: Assume that the internal energy change is entirely absorbed by the liquid resulting is a mass of vapor. Use the Debye model to calculate the change in internal energy, (Fe) = 460 K; hfg(He@ 4 K)=21 kJ/kg; hfg (N₂ @ 80 K=200 kJ/kg)).
Expert Answer:
Answer rating: 100% (QA)
Mass of iron magnet 2000 kg Initial temperature 300 K Final temperature for liquid nitrogen cooling ... View the full answer

Related Book For 

Fundamentals of Thermodynamics
ISBN: 978-0471152323
6th edition
Authors: Richard E. Sonntag, Claus Borgnakke, Gordon J. Van Wylen
Posted Date:
Students also viewed these finance questions
-
Helium in a steel tank is at 250 kPa, 300 K with a volume of 0.1 m3. It is used to fill a balloon. When the tank pressure drops to 150 kPa the flow of helium stops by itself, if all the helium still...
-
In a physics lab, a student accidentally drops a 25.0-g brass washer into an open dewar of liquid nitrogen at 77.2 K. How much liquid nitrogen boils away as the washer cools from 293 K to 77.2 K? The...
-
Someone offered the investment options to Hendry on January 1, 2023: 1. Hendry has to save up to 5 times the initial deposit of US $ 150,000/year. 2. The savings cannot be taken for 20 years until...
-
Suppose m and r are integers. Write a Java expression for mr2 to obtain a floating point result.
-
Janis has demand for 40,000 desks p.a. and the purchase price of each desk is $25. There are ordering costs of $20 for each order placed. Inventory holding costs amount to 10% p.a. of inventory...
-
White Brothers manufactures conveyor belts. Early in May 2011, White Brothers constructed its own building at a materials, labor, and overhead cost of $970,000. White Brothers also paid for architect...
-
An company has sales of $50 million growing at 25% YoY with EBITDA margins at 20%. It secures a JV in Year 3 with additional business of $20 million at EBITDA of 12.5% which linearly scales up to $50...
-
Howell Corporation has a job order cost system. The following debits (credits) appeared in Work in Process for the month of July: July 1, balance....... July 31, direct materials.. July 31, direct...
-
Segment the wind-generated power industry in a way that shows where Windspire Energy, the subject of the You Be the VC 1.1 feature, is located in the industry. How does Windspires awareness of the...
-
Find the inverse of the matrix A, if it exists. Show your work on scratch sheet. 2-31 Edit Format Table 12pt Paragraph BIUA 2 Tv 202 :
-
How has the firm Goodyear's corporate-level strategy evolved over time? Explain
-
MUNOZ COMPANY Balance sheet Assets Cash $ 14,800 Marketable securities 7,860 Accounts receivable Inventory Property and equipment Accumulated depreciation Total assets Liabilities and Stockholders'...
-
How can organizations proactively address the ethical and moral dilemmas inherent in the pursuit of organizational development initiatives, balancing the imperative for growth and profitability with...
-
Marigold Corporation had 8 6 0 0 0 0 shares of common stock outstanding during the year. Marigold declared and paid cash dividends of $ 4 7 3 0 0 0 on the common stock and $ 3 3 0 0 0 0 on the...
-
At the end of 2 0 X 9 , SpendCo has accounts receivable of $ 7 0 , 0 0 0 , of which it estimates 1 0 % will be bad debt. Allowance for Doubtful Accounts has a debit balance of $ 4 , 0 0 0 . A ) What...
-
In R, two non-parallel, non-intersecting lines are called skew lines. (a) (3 marks) Show that the lines x-3 2 =4-y= 2-1 3 L : L: x=1-3-y_254 2 5 are skew lines. (b) (3 marks) Determine the scalar...
-
QUESTION 2 The CEO of Farisha Hijab Sdn Bhd insisted on further investigation to be carried out that he also required Mr Muaz to conduct the analysis of variance for the material and labour of the...
-
Do Problem 11.26 with ammonia as the working fluid. A supply of geothermal hot water is to be used as the energy source in an ideal Rankine cycle, with R-134a as the cycle working fluid. Saturated...
-
Consider an ideal Stirling-cycle engine in which the state at the beginning of the isothermal compression process is 100 kPa, 25C, the compression ratio is 6, and the maximum temperature in the cycle...
-
Ethane is burned with 150% theoretical air in a gas turbine combustor. The products exiting consist of a mixture of CO2, H2O, O2, N2, and NO in chemical equilibrium at 1800 K, 1 MPa. Determine the...
-
Consider the MOSFET feedback amplifier shown in Figure P12.39. The transistor parameters are \(V_{T N}=0.5 \mathrm{~V}, K_{n}=0.5 \mathrm{~mA} / \mathrm{V}^{2}\), and \(\lambda=0\). Determine the...
-
The transistor parameters for the circuit in Figure P12.42 are: \(h_{F E}=50\), \(V_{B E}(\mathrm{on})=0.7 \mathrm{~V}\), and \(V_{A}=\infty\). Using nodal analysis, determine the closedloop...
-
The parameters of the basic source-follower circuit in Figure P12.41 are \(R_{S}=1.5 \mathrm{k} \Omega, V_{T N}=1.2 \mathrm{~V}\), and \(\lambda=0\). Assume the transistor is biased at \(I_{D Q}=1.2...

Study smarter with the SolutionInn App


