Question: Calculate the loganthm for 7 X = (2.81)
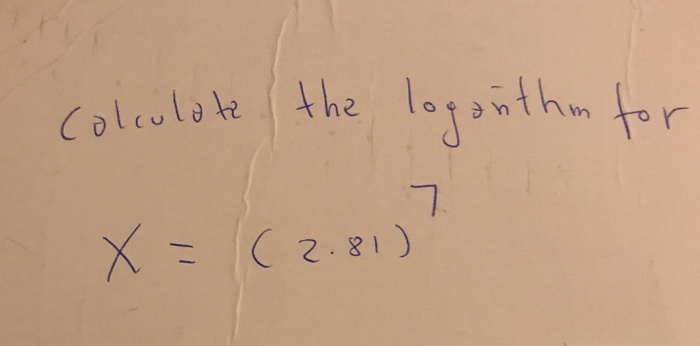
Calculate the loganthm for 7 X = (2.81)
Step by Step Solution
3.45 Rating (148 Votes )
There are 3 Steps involved in it
Solution Given X 281 We have to find it... View full answer

Get step-by-step solutions from verified subject matter experts


