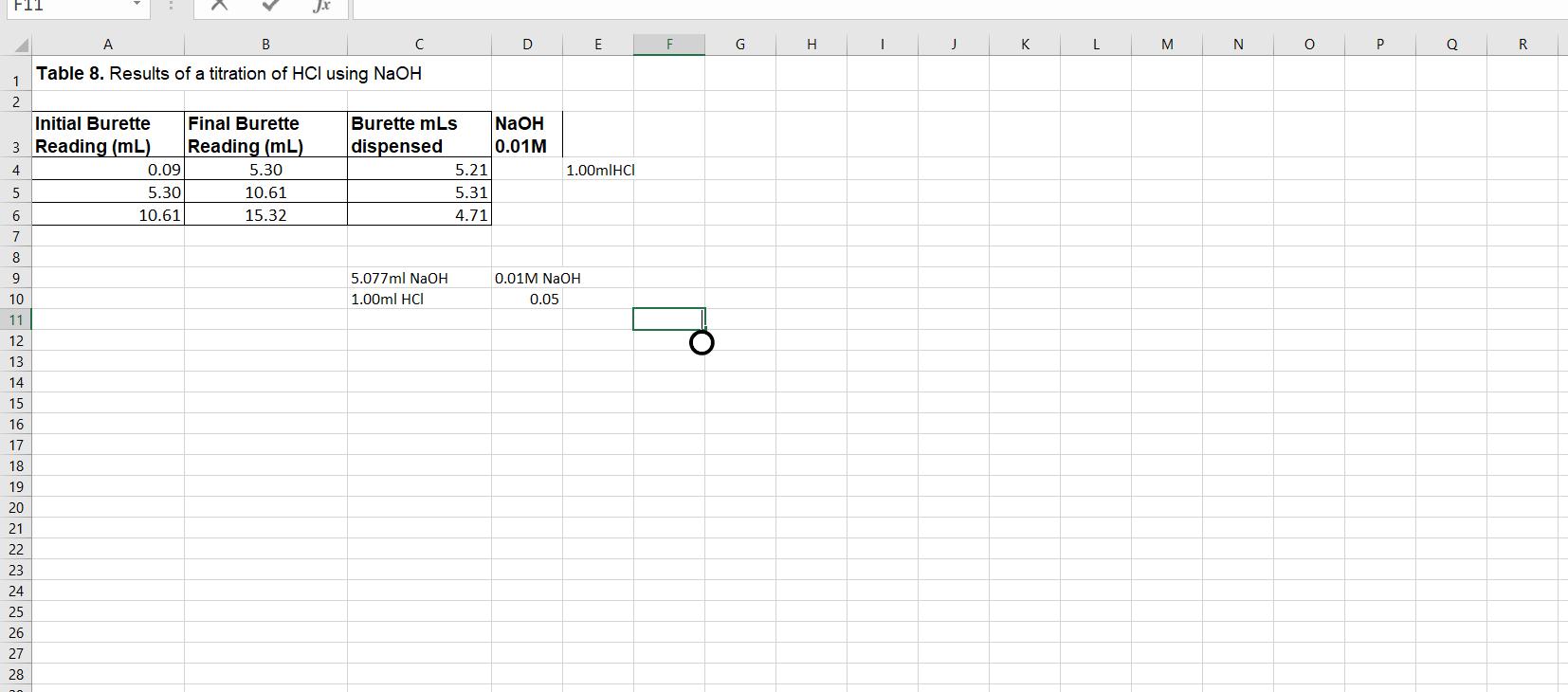
Calculate the molarity of the HCl solution used in the titration in Procedure 8. You should calculate
Fantastic news! We've Found the answer you've been seeking!
Question:
Calculate the molarity of the HCl solution used in the titration in Procedure 8. You should calculate the molarity for each of your three measurements, and then calculate the mean molarity (M) and the standard error. You should present you molarity in this form: XXX ± XXX M (± SEM) Show your work. Be sure to consider significant figures in your calculations. The equation for the reaction is:
NaOH + HCl → NaCl + H2O
Hint: You know the concentration of the NaOH (0.01 mole/ L) and you measured the number of mL of this solution used to neutralize the HCl.
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:




