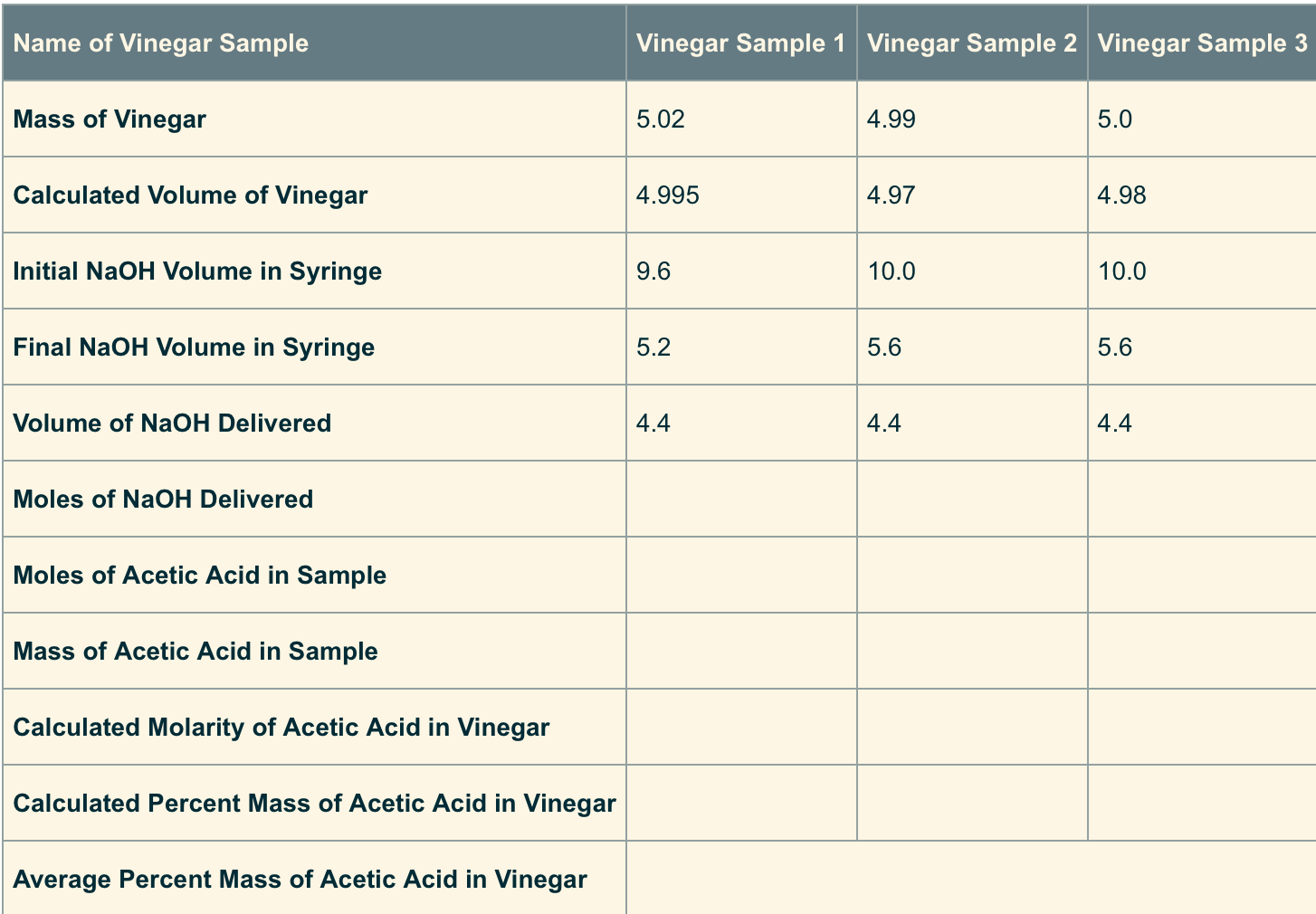
Calculate the moles of NaOHdelivered by using the volume of NaOH and themolarity of NaOH. (The volume
Fantastic news! We've Found the answer you've been seeking!
Question:
- Calculate the moles of NaOHdelivered by using the volume of NaOH and themolarity of NaOH. (The volume needs to be in liters and theconcentration of the NaOH is 1 M.)
- The moles of acetic acid insample is equal to the moles of NaOH deliveredbecause the acid has one H+ and the base has oneOH-. So it is a 1 to 1 ratio.
- The mass of acetic acid insample can be found by converting moles of aceticacid (HC2H3O2) to grams of aceticacid. (You may need to go back and review how to do molecalculations from unit 3.)
- The calculated molarity of acetic acid in thesampled vinegar is found using the molarity equation:moles of solute divided by liters of total solution.
- The calculated percent mass of acetic acid insampled vinegar is found by taking the mass of aceticacid in the sample divided by the mass of the vinegar andmultiplying by 100.
- Once all three trials are done, you can calculatethe average percent mass of acetic acid in sampledvinegar.
Related Book For 

Operations Management Creating Value Along the Supply Chain
ISBN: 978-0470525906
7th Edition
Authors: Roberta S. Russell, Bernard W. Taylor
Posted Date:




