Calculate the pH of a solution in which [H3O'] = 6.85x103 M. Give your answer to...
Fantastic news! We've Found the answer you've been seeking!
Question:
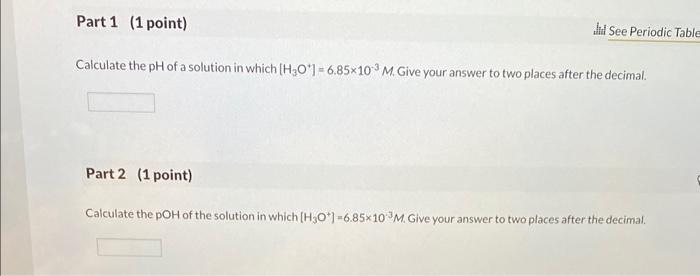

Transcribed Image Text:
Calculate the pH of a solution in which [H3O'] = 6.85x103 M. Give your answer to two places after the decimal. Part 2 (1 point) Jd See Periodic Table Calculate the pOH of the solution in which [H3O*] -6.85x10M. Give your answer to two places after the decimal. Part 3 (1 point) If 100. mL of the solution in part 1 is concentrated by evaporation to give a final volume of 1.00 mL, what is the change in pl Choose one: pH decreases by 2. pH increases by 2. pH decreases by 10. pH increases by 10. Calculate the pH of a solution in which [H3O'] = 6.85x103 M. Give your answer to two places after the decimal. Part 2 (1 point) Jd See Periodic Table Calculate the pOH of the solution in which [H3O*] -6.85x10M. Give your answer to two places after the decimal. Part 3 (1 point) If 100. mL of the solution in part 1 is concentrated by evaporation to give a final volume of 1.00 mL, what is the change in pl Choose one: pH decreases by 2. pH increases by 2. pH decreases by 10. pH increases by 10.
Expert Answer:
Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:
Students also viewed these chemistry questions
-
Calculate the pH of a solution in which the concentration of piperdine is (a) 1.00 10-1 M. (b) 1.00 10-2 M. (c) 1.00 10-4 M.
-
Calculate the pH of a solution made up from 2.0 g of potassium hydroxide dissolved in 115 mL of 0.19 M perchloric acid. Assume the change in volume due to adding potassium hydroxide is negligible.
-
What is the pH of a solution in which 15 mL of 0.10 M NaOH has been added to 25 mL of 0.10 M HCl?
-
The administrator of Hope Hospital has been asked to perform an activity analysis of the emergency room (ER). The ER activities include cost of quality and other patient care activities. The lab...
-
1. Using the categories discussed in the chapter, what type of negotiable instrument was the note that Durbin signed (an order to pay or a promise to pay)? Explain. 2. Suppose that the note did not...
-
To what extent does the adoption of agile frameworks engender a reconfiguration of traditional project management paradigms, catalyzing a departure from rigid, top-down approaches towards adaptive,...
-
What is the difference between a letter of transmittal and letter of authorization?
-
Starbucks is hoping to make use of its excess restaurant capacity in the evenings by experimenting with selling beer and wine. It speculates that the only additional costs are hiring more of the same...
-
A simple loan with a present value of $5500 will be paid off in 5 years. If the interest rate is 7% what will the future payment be?
-
On January 1, 2020, Xiamen Company made amendments to its defined benefit pension plan that resulted in 62,800 yuan of past service cost. The plan has 5,130 active employees with an average expected...
-
Identify and describe the purpose of a needs assessment?
-
What are the disadvantages of separating financial activities into different firms in an effort to avoid conflicts of interest?
-
How can conflicts of interest make financial markets less efficient?
-
In the July 2017 FOMC meeting, governors and voting presidents of the Federal Reserve System agreed not to increase the federal funds rate target, but somewhat let the markets know that there could...
-
Refer to Problem 22. Now you believe the dealer knows more about the car than you do. How much are you willing to pay? Why? How can this asymmetric information problem be resolved in a competitive...
-
This case study shows a typical situation in which management accounting can be helpful. Read the case study now but only attempt the discussion points after you have finished studying the chapter....
-
A toggle mechanism is shown in Figure 1. With link 2 rotating at constant angular velocity, determine velocity of the slider block, 03, 004 and 5 by drawing a velocity polygon. Also determine the...
-
Find the work done in pumping all the oil (density S = 50 pounds per cubic foot) over the edge of a cylindrical tank that stands on one of its bases. Assume that the radius of the base is 4 feet, the...
-
A 50.0-mL portion of a solution containing 0.200 g of BaCl2 2H2O is mixed with 50.0 mL of a solution containing 0.300 g of NaIO3. Assume that the solubility of Ba(IO3)2 in water is negligibly small...
-
Iron (III) forms a complex with thiocyanate ion that has the formula Fe(SCN)2+. The complex has an absorption maximum at 580 nm. A specimen of well water was assayed according to the scheme below....
-
Show that one gram is one mole of unified atomic mass units.
-
Input information from the SBA Personal Financial Statements given to Sharptop Bank into a spreadsheet. a. Separate personal items (e.g., residence) from business items (e.g., inventory). b....
-
Compare your recalculated ending inventory to that given to Sharptop Bank and to Southern Appalachian Insurance. Is either estimated ending inventory materially different from that reported on the...
-
What accuracy certifications on the tax return and bank financial statement address providing materially inaccurate information?

Study smarter with the SolutionInn App



