What volume in liters would this gas occupy at STP? Express your answer to four significant...
Fantastic news! We've Found the answer you've been seeking!
Question:
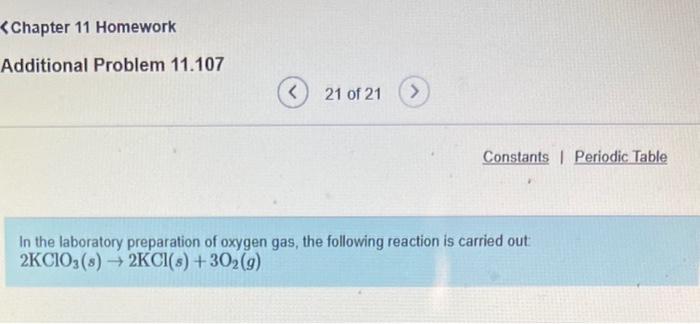
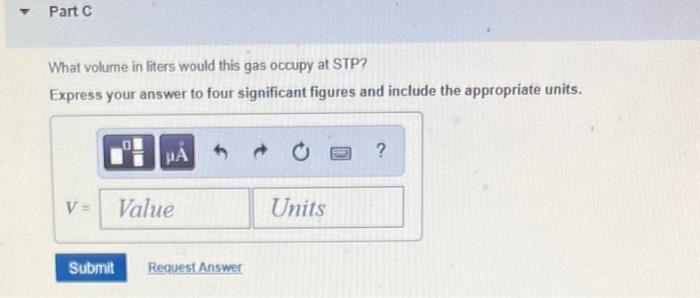
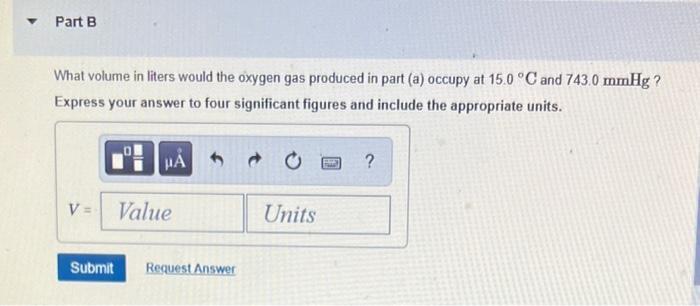
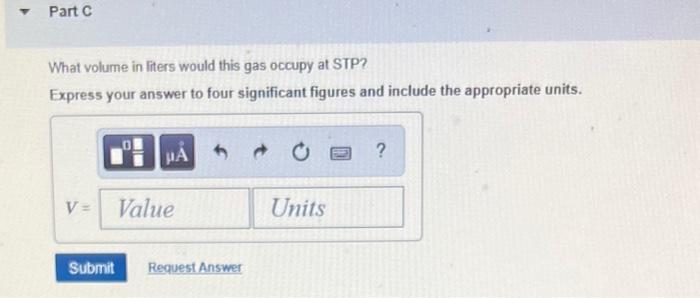
Transcribed Image Text:
What volume in liters would this gas occupy at STP? Express your answer to four significant figures and include the appropriate units. HA V = Value Submit Request Answer Units ? Part B What volume in liters would the oxygen gas produced in part (a) occupy at 15.0 C and 743.0 mmHg? Express your answer to four significant figures and include the appropriate units. A V = Value Submit Request Answer Units ? Part C What volume in liters would this gas occupy at STP? Express your answer to four significant figures and include the appropriate units. HA V = Value Submit Request Answer Units ? What volume in liters would this gas occupy at STP? Express your answer to four significant figures and include the appropriate units. HA V = Value Submit Request Answer Units ? Part B What volume in liters would the oxygen gas produced in part (a) occupy at 15.0 C and 743.0 mmHg? Express your answer to four significant figures and include the appropriate units. A V = Value Submit Request Answer Units ? Part C What volume in liters would this gas occupy at STP? Express your answer to four significant figures and include the appropriate units. HA V = Value Submit Request Answer Units ?
Expert Answer:
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Posted Date:
Students also viewed these chemistry questions
-
What volume in Liters would a 0.871 gram sample of air occupy if the density of air is 1.29 g/L?
-
Using physical constants accurate to four significant figures (see inside front cover), verify the numerical values of the Bohr radius a 0 and the hydrogen ground-state energy E 1 .
-
Perform each calculation and limit each answer to four significant figures. a. 18,900 76.33 ( 0.00336 = ? b. 0.77604 ( 76,003 8.888 = ?
-
Based on the following information, calculate the sustainable growth rate for Kaleb's Welding Supply: Profit margin = 7.5% Capital intensity ratio = .65 Debt-equity ratio = .60 Net income = $67,000...
-
What reasonable steps could Auto-Owners have taken to prevent such internal fraud? Kenneth Wulf worked in the claims department of Auto- Owners Insurance Company for ten years. When the department...
-
The physics of a solenoid and the electromagnet. The coils of a wire in a solenoid are arranged as shown below. a. Draw the magnetic field line pattern for this arrangement of current. b. A compass...
-
Why is the executive summary an important part of the report?
-
The beginning inventory for Dunne Co. and data on purchases and sales for a three-month period are shown in Problem 6-1B. In Problem 6-1B The beginning inventory of merchandise at Dunne Co. and data...
-
Grossnickle Corporation issued 20-year, noncallable, 9.7% annual coupon bonds at their par value of $1,000 one year ago. Today, the market interest rate on these bonds is 7.4%. What is the current...
-
1H NMR spectra for two haloalkanes are shown below. Propose structures for these compounds that are consistent with the spectra. (a) C5H11C1, spectrum A; (b) C4H8Br2, spectrum B. H NMR 9 H 2 H...
-
A double replacement reaction between lead(IV) nitrate and sodium chloride will produce lead(IV) chloride and sodium nitrate. If 1.02 moles of lead(IV) nitrate is reacted, how many grams of sodium...
-
How can poorly designed compensation schemes in financial services firms lead to conflicts of interest?
-
Give two examples of conflicts of interest that do not seem to have been exploited and thus have not led to a reduction of reliable information in the financial markets.
-
What are reputational rents, and how are they significant?
-
What are the advantages and disadvantages of governmental provision of information as a solution to the problems created by conflicts of interest?
-
If the price of the Treasury bond is 115 at expiration, is the option at the money, in the money or out of the money? Determine the premium if the profit equals $8,000.
-
2. [4 marks]. Let A be a given n x n matrix, where n is a positive integer. Let S be the set of all n x n matrices B having the property that AB = BA. Let us prove that S is a subspace. (a) [2...
-
A test car is driven a fixed distance of n miles along a straight highway. (Here n Z+.) The car travels at one mile per hour for the first mile, two miles per hour for the second mile, four miles...
-
This question is about the reaction: N 2 (g) + 3H 2 (g) 2NH 3 (g) H r = 92 kJ mol 1 120.0mol of hydrogen gas are mixed with 40.0mol of nitrogen gas then pressurised. The mixture of gases is passed...
-
a. Identify the acid and the base on the right-hand side of these equilibria. i. HClO 2 + HCOOH ClO 2 + HCOOH 2 + ii. H 2 S + H 2 O HS + H 3 O + b. Identify the acid on the right-hand side of...
-
Show the mechanism, including appropriate curly arrows, for the hydrolysis of 1- chloropropane, CH 3 CH 2 CH 2 Cl, by alkali.
-
Henry Ford, founder of Ford Motor Company, is quoted as saying that customers could choose a car in any color as long as it was black. Things have come a long way since that timewhen customization...
-
Not long ago, New England Confectionery Company, or Necco for short, marked the production of its one trillionth candy wafer. The humble roots of Necco, the country's oldest continuously operating...
-
What is utility?

Study smarter with the SolutionInn App



