Calculate the phase diagram of Ge-Si binary system assuming that Ge and Si form ideal solutions...
Fantastic news! We've Found the answer you've been seeking!
Question:
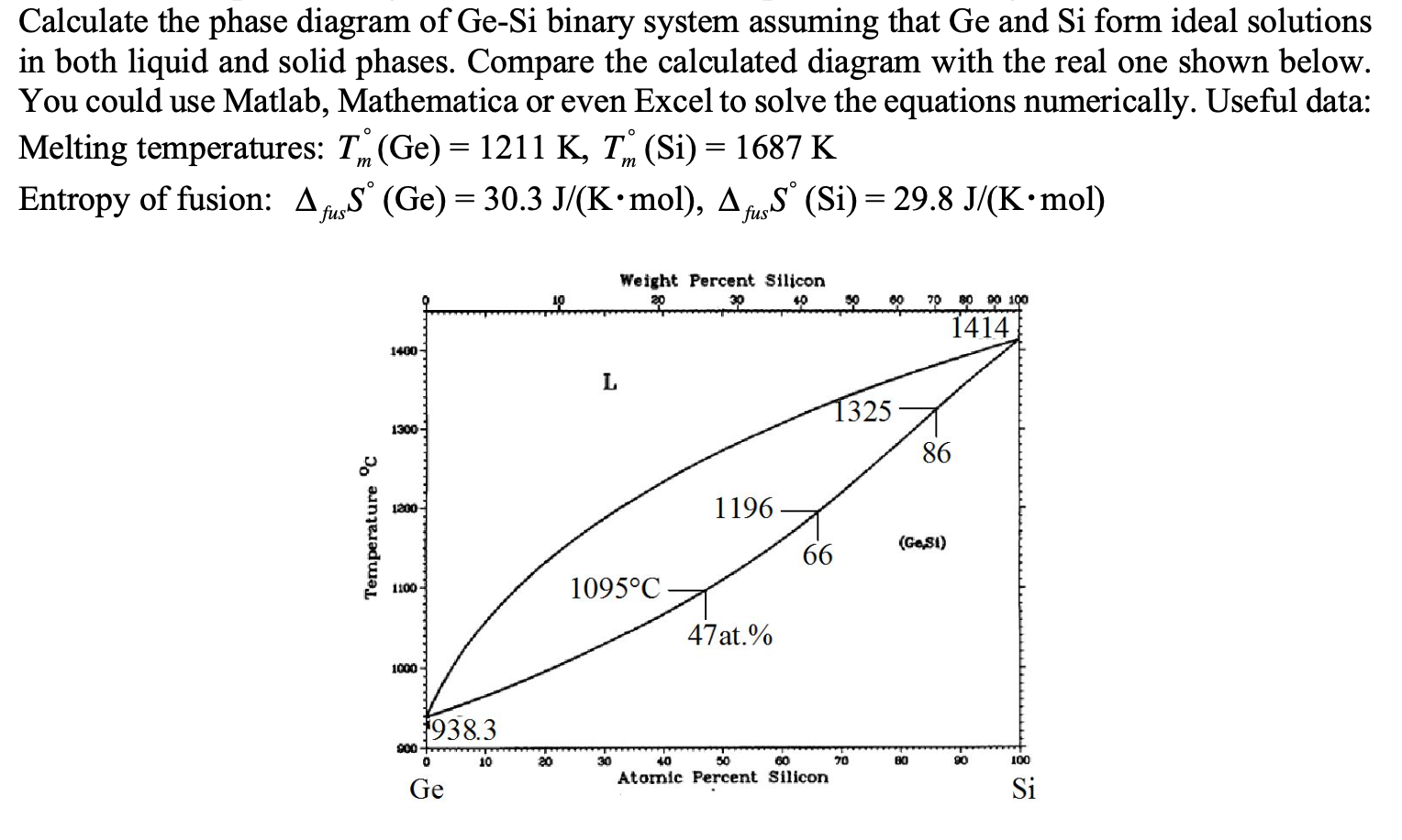
Transcribed Image Text:
Calculate the phase diagram of Ge-Si binary system assuming that Ge and Si form ideal solutions in both liquid and solid phases. Compare the calculated diagram with the real one shown below. You could use Matlab, Mathematica or even Excel to solve the equations numerically. Useful data: Melting temperatures: T (Ge) = 1211 K, T (Si) = 1687 K O m m Entropy of fusion: AS (Ge)= 30.3 J/(K·mol), AS (Si) = 29.8 J/(K.mol) Temperature ºc 1400- 1300- 1200- 1100- 1000 900 1938.3 0 Ge 10 10 20 L Weight Percent Silicon 20 30 40 1095°C- 30 40 1196 47at.% 1325 66 50 60 Atomic Percent Silicon 70 70 80 90 100 1414 80 86 (Ge,S1) 90 100 Si Calculate the phase diagram of Ge-Si binary system assuming that Ge and Si form ideal solutions in both liquid and solid phases. Compare the calculated diagram with the real one shown below. You could use Matlab, Mathematica or even Excel to solve the equations numerically. Useful data: Melting temperatures: T (Ge) = 1211 K, T (Si) = 1687 K O m m Entropy of fusion: AS (Ge)= 30.3 J/(K·mol), AS (Si) = 29.8 J/(K.mol) Temperature ºc 1400- 1300- 1200- 1100- 1000 900 1938.3 0 Ge 10 10 20 L Weight Percent Silicon 20 30 40 1095°C- 30 40 1196 47at.% 1325 66 50 60 Atomic Percent Silicon 70 70 80 90 100 1414 80 86 (Ge,S1) 90 100 Si
Expert Answer:
Answer rating: 100% (QA)
Temperature c 1400 1300 1200 1100 1000 9383 008 Ge 10 10 34 20 L Weight Percent Silicon ... View the full answer

Related Book For 

Essentials of Materials Science and Engineering
ISBN: 978-1111576851
3rd edition
Authors: Donald R. Askeland, Wendelin J. Wright
Posted Date:
Students also viewed these chemical engineering questions
-
The phase diagram of sulfur is shown here. (a) How many triple points are there? (b) Monoclinic and rhombic are two allotropes of sulfur. Which is more stable under atmospheric conditions? (c)...
-
The phase diagram of a hypothetical substance is (a) Estimate the normal boiling point and freezing point of the substance. (b) What is the physical state of the substance under the following...
-
The phase diagram of liquid 3 He 4 He mixtures in Figure shows that the solubility of 3 He in 4 He remains finite (about 6 pct) as 0. Similarly, the Pb-Sn phase diagram of Figure shows a finite...
-
Write a critical review paper on the topic of financial management in the broad sense.
-
Many organizations, especially multinational corporations, must consolidate reports to ensure smooth operations. These reports include currency, measurements, and dates. How would you help them...
-
Consider a steel sphere (SG = 7.85) of diameter 2 cm, dropped from rest in water at 20C. Assume a constant drag coefficient CD = 0.47. Accounting for the spheres hydrodynamic mass, estimate (a) its...
-
While the electromagnetic interaction between the photons and the charged electrons and positrons kept them in equilibrium with each other during the early universe, show that the direct...
-
The stockholders equity accounts of G.K. Chesterton Company have the following balances on December 31, 2008. Common stock, $10 par, 300,000 shares issued and outstanding ... $3,000,000 Paid-in...
-
Max Labs Inc. current portfolio of products has a 10% IRR and a 10% standard deviation. The correlation coefficients with the firm portfolio are 0.5 (r=0.5) for both, Dog Treats and Dog Food; zero...
-
1. Pick a futures that you are interested in. ie, a commodity (iron, barley, etc), financial futures (index), etc. 2. State the market your research is based on ie, ASX, CBOE, etc. 3. Put in a price...
-
sorted function and dict inbuilt type should not be used . should be in python A WalkerPersonality is a class that implements two methods: add_mountain , which allows a Walker to note that mountain...
-
Zeneda Inc. is estimating its WACC. You know the following information about the company's capital structure: Bonds (Bonds are the only debt) 10 years maturity - 7% coupon, semiannually payable...
-
Define customer service (cite the source for paraphrasing). What are the key factors - what determines exceptional customer service, acceptable customer service, and poor customer service? How does...
-
a) how fast is the water in the river flowing with respect to the ground in m/s ? b) what is the speed of the swimmer with respect to a friend at rest on the ground in m/s ? An athlete crosses a 23 m...
-
Determine if the following systems are time-invariant, linear, causal, and/or memoryless. dy(t) a) + 5y(t) = 4x(t) dt dy(t) b) dt c) y[n] + 2y[n- 3] = x[n + 1] d) y(t) = 3sin (x(t)) + 4ty(t) = x(t)...
-
If McDonald's seeks to maintain cost efficiency and continuity in its communication programs, the company has likely adopted a ____________ advertising department. Categorized Long-term Global...
-
A 100 mm diameter orifice under a head of 4.88 m discharges 8900 N if water in 32.6 sec. The trajectory was determined by measuring 4.76 m horizontal distance traveled by the water jet for a drop of...
-
The graph of the sequence whose general term is an = n - 1 is which of the following? [8.1] A. B. TITTT 3-2-1 23.45 2.3.4
-
We would like to produce a lightweight epoxy part to provide thermal insulation. We have available hollow glass beads for which the outside diameter is 1/16 in. and the wall thickness is 0.001 in....
-
Molten copper at atmospheric pressure contains 0.01 wt% oxygen. The molten copper is placed in a chamber that is pumped down to 1 Pa to remove gas from the melt prior to pouring into the mold....
-
(a) Determine the critical nucleus size r* for homogeneous nucleation for precipitation of phase in a matrix of phase . (b) Plot the total free energy change G as a function of the radius of the...
-
How is the legal infrastructure of a country significant for business?
-
What is legal liability?
-
We have a parliament to pass laws, a government to administer laws, and a police department to enforce laws. Ironically, these potent instruments for the restriction of liberty are necessary for the...

Study smarter with the SolutionInn App


