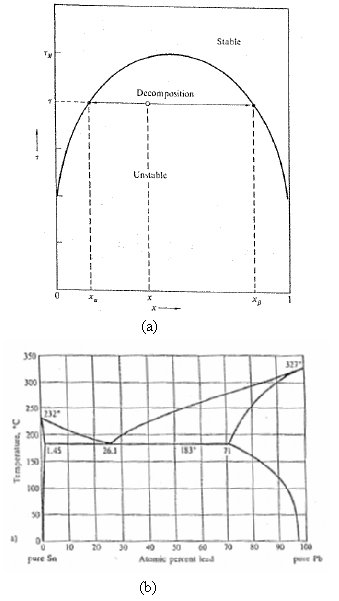
The phase diagram of liquid 3 He 4 He mixtures in Figure shows that the solubility of
Question:
The phase diagram of liquid 3He– 4He mixtures in Figure shows that the solubility of 3He in 4He remains finite (about 6 pct) as τ → 0. Similarly, the Pb-Sn phase diagram of Figure shows a finite residual solubility of Pb in solid Sn with decreasing τ. What do such finite residual solubilities imply about the form of the function u(x)?
Transcribed Image Text:
Stabte Decomposition Unstable (a) 350 327 300 250 232 200 L.4S 190 I83 26.1 71 00 a) 20 80 10 20 30 30 90 100 pure Se Atomis peneni lead (b) Temperature, "C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
The curve ux must have a shape qualitatively ...View the full answer

Answered By

Lilian Nyambura
Hi, am Lilian Nyambura, With extensive experience in the writing industry, I am the best fit for your writing projects. I am currently pursuing a B.A. in Business Administration. With over 5 years of experience, I can comfortably say I am good in article writing, editing and proofreading, academic writing, resumes and cover letters. I have good command over English grammar, English Basic Skills, English Spelling, English Vocabulary, U.S. English Sentence Structure, U.K. or U.S. English Punctuation and other grammar related topics. Let me help you with all your essays, assignments, projects, dissertations, online exams and other related tasks. Quality is my goal.
4.80+
378+ Reviews
750+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Solid State questions
-
The phase diagram of sulfur is shown here. (a) How many triple points are there? (b) Monoclinic and rhombic are two allotropes of sulfur. Which is more stable under atmospheric conditions? (c)...
-
The phase diagram of a hypothetical substance is (a) Estimate the normal boiling point and freezing point of the substance. (b) What is the physical state of the substance under the following...
-
Use graph paper and sketch the phase diagram of oxygen, O2, from the following information: normal melting point, 218C; normal boiling point, 183C; triple point, 219C, 1.10 mmHg; critical point,...
-
Southern Stitches is a local casual clothing shop that makes a variety of t-shirt styles. Below is some actual vs. budget information for Southern's t-shirts for the month of May. Assume no...
-
This problem illustrates a deceptive way of quoting interest rates called add-on interest. Imagine that you see an advertisement for Crazy Judy's Stereo City that reads something like this: "$1,000...
-
Hailey Corp. pays a constant $9.45 dividend on its stock. The company will maintain this dividend for the next 13 years and will then cease paying dividends forever. If the required return on this...
-
Consider the methanol oxidation data in Table B.20. Perform a thorough analysis of these data. What conclusions do you draw from this analysis? x x2 X3 X4 y 0 454 8.8 3.90 1.30 1.1 0 474 8.2 3.68...
-
The 2010 oil spill in the Gulf of Mexico caused the oil firm BP and the U. S. government to greatly increase purchases of boat services, various oil-absorbing materials, and other goods and services...
-
6. Three charges qi, q2 and qs are arranged as shown. The charges are 9.5 C, -1.8C and -5.8 C respectively. What is the force acting on q? (6 marks) 0. 91 0.30 m 0.50 m 92 0.40 m 93
-
On January 1, 2014, Dunbar Corporation, an 85%-owned subsidiary of Garfield Industries, received $48,055 for $50,000 of 8%, 5-year bonds it issued when the market rate was 9%. When Garfield...
-
Show that the chemical potentials A and B of the two atomic species A and B of an equilibrium two phase mixture are given by the intercepts of the two-point tangent in Figure with the vertical...
-
Let B be an impurity in A, with X < < 1. In this limit the non-mixing parts of the free energy can be expressed as linear functions of x, as f0(x) = f0(0) + xf0(0), for both liquid and solid phase....
-
Your task is to prepare styrene by one of the following reactions. Which reaction would you choose to give the better yield of styrene? Explain your answer. (1) Styrene Or (2) Styrene Br KOH EtOH, ...
-
Brazil, one of the worlds largest emerging markets with the ninth largest economy, has a population of 208 million and GDP of $1.8 trillion.97 In 2014, a federal police superintendent in Brazil began...
-
The chapter describes six examples of cross-border mergers: can you identify the source of long-term value creation in each case?
-
What are the main difficulties that confront expatriate managers when they are assigned to an emerging country?
-
Why can global pricing lead to sub-optimization?
-
Make a CAGE analysis of Morocco, Romania and South Korea. What conclusions do you draw from the point of view of a South Korean firm willing to invest in an assembly plant in one of these two...
-
What is the deliverable that is created once requirements analysis is completed? Why is this deliverable needed, and what does it include? Who are the audience and/or users of this deliverable, and...
-
Write the given system without the use of matrices. D) - ()- d (x sin t + 8 (2+ 1)
-
Write the SQL code for the following: Order the reservations by class date and then by class ID. Display all fields. a. SELECT * FROM Reservations ORDER BY ClassDate, ClassID; b. DISPLAY ALL FROM...
-
One mole of an ideal gas is contained in a cylinder with a movable piston. The initial pressure, volume, and temperature are Pi, Vi, and Ti, respectively. Find the work done on the gas for the...
-
One mole of an ideal gas, initially at 300 K, is cooled at constant volume so that the final pressure is one fourth of the initial pressure. Then the gas expands at constant pressure until it reaches...
-
Continue the analysis of Problem 60 in Chapter 19. Following a collision between a large spacecraft and an asteroid, a copper disk of radius 28.0 m and thickness 1.20 m, at a temperature of 850C, is...
-
Can we justify the acquisition of Peloton Interactive Inc using NPV assuming the initial investment is 10% of the total market value of the company, 5% constant growth rate for the next three years...
-
Mary would like to save $10,000 at the end of 5 years for a future down payment on a car. How much should she deposit at the end of each month in a savings account that pays 1.2%/a, compounded...
-
AA stock price is $45/share. The stock is expected to pay dividends $2.50 in the coming year. After reviewing all information, you concluded: the price one year from now will be $50/share and you...

Study smarter with the SolutionInn App


