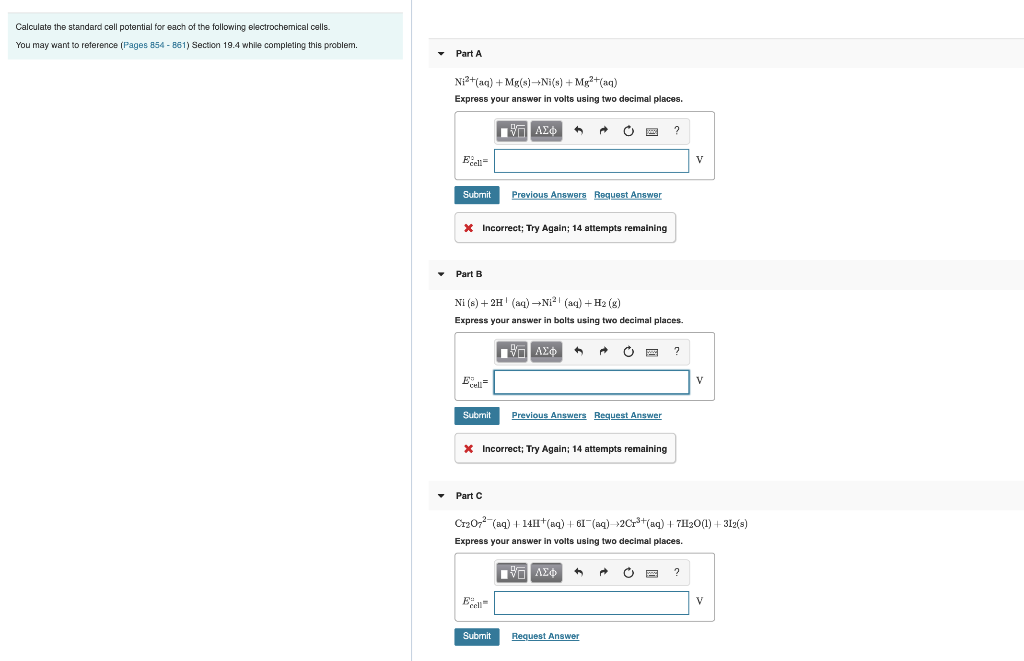
Calculate the standard cell potential for each of the following electrochemical cells. You may want to...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Calculate the standard cell potential for each of the following electrochemical cells. You may want to reference (Pages 854-861) Section 19.4 while completing this problem. Part A Ni²+ (aq) + Mg(s)→Ni(s) +Mg²+(aq) Express your answer in volts using two decimal places. Excell Submit Part B * Incorrect; Try Again; 14 attempts remaining Ecell Submit 17 ΑΣΦ Ni (s) + 2H¹ (aq) →N₁²¹ (aq) + H₂(g) Express your answer in bolts using two decimal places. ▾ Part C ← Previous Answers Request Answer Ecell Submit |-- ΑΣΦ VO * Incorrect; Try Again; 14 attempts remaining ^ → Previous Answers Request Answer ? ← Cr₂O7² (aq) + 14H+ (aq) + 61¯(aq)→2Cr³+ (aq) +7H₂O(1)+312(s) Express your answer in volts using two decimal places. [35] ΑΣΦ Request Answer ? V 2 V V Calculate the standard cell potential for each of the following electrochemical cells. You may want to reference (Pages 854-861) Section 19.4 while completing this problem. Part A Ni²+ (aq) + Mg(s)→Ni(s) +Mg²+(aq) Express your answer in volts using two decimal places. Excell Submit Part B * Incorrect; Try Again; 14 attempts remaining Ecell Submit 17 ΑΣΦ Ni (s) + 2H¹ (aq) →N₁²¹ (aq) + H₂(g) Express your answer in bolts using two decimal places. ▾ Part C ← Previous Answers Request Answer Ecell Submit |-- ΑΣΦ VO * Incorrect; Try Again; 14 attempts remaining ^ → Previous Answers Request Answer ? ← Cr₂O7² (aq) + 14H+ (aq) + 61¯(aq)→2Cr³+ (aq) +7H₂O(1)+312(s) Express your answer in volts using two decimal places. [35] ΑΣΦ Request Answer ? V 2 V V
Expert Answer:
Answer rating: 100% (QA)
Answer Ans A Ni aq Mgs E cell Cathode Ni s Mg 2 aq An... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the standard cell potential for each of the followingelectrochemical cells. Part A Ni2+( a q )+Mg( s )?Ni( s )+Mg2+( a q ) Express your answer using two decimal places. E ?cell = _____ V...
-
while conducting your analysis you may want to consider and identify the various design elements that are present in the excerpt (ie, use of alignment, chunking, use of color, etc) how effective are...
-
Calculate the yield to maturity for each of the following bonds. a. A 20-year bond with a coupon of $100, principal payment at maturity of $2,000, and a current price of $2,000. b. A 5-year bond with...
-
Rocket Science A rocket has total mass Mi = 360 kg, including 330 kg of fuel and oxidizer. In interstellar space it starts from rest, turns on its engine at time t = 0, and puts out exhaust with...
-
The Occupational Safety and Health Act empower agents of the Secretary of Labor to search the work area of any employment facility within the acts jurisdiction. No search warrant or other process is...
-
The financial accounting balance sheet for Zane, a retail sales corporation, as of the end of the current year is as follows: Using the accrual method Zane reports the following financial accounting...
-
Hulus Hybrids has the following information for the years ending January 31 2010 and 2009: Requirements 1. Compute the rate of inventory turnover for Hulus Hybrids for the years ended January 31,...
-
A statement of financial affairs created for an insolvent corporation that is beginning the process of liquidation discloses the following data (assets are shown at net realizable values): Assets...
-
Classic Clothing, Inc., is a manufacturer of winter clothes. It has a knitting department and a finishing department. This exercise focuses on the finishing department. Direct materials are added at...
-
23. The force between the plates of a parallel plate capacitor of capacitance C and distance of separation of the plates d with a potential difference V between the plates is :- (1) CV/2d (3) Cv/d...
-
Explain the jurisdictional role of UIFSA in support cases.
-
What is a Problem?
-
What kinds of marriage restriction laws are in effect today?
-
List some nonfunctional requirement rules.
-
How did the Puritans view divorce?
-
The company name is iTechskills. Website : https://itechskills.ca/ Please elaborate or explain : Company highlights: watershed event, summarized history, or any description of the company's reason...
-
Suppose the spot and six-month forward rates on the Norwegian krone are Kr 5.78 and Kr 5.86, respectively. The annual risk-free rate in the United States is 3.8 percent, and the annual risk-free rate...
-
Liquid nitrogen an be used for the quick freezing of foods. The liquid boils at 196oC. What is this temperature in degrees Fahrenheit?
-
What are the different kinds of particles in the atoms nucleus? Compare their properties with each other and with those of an electron.
-
A student gently drops an object weighing 15.8 g into an open vessel that is full of ethanol, so that a volume of ethanol spills out equal to the volume of the object. The experimenter now finds that...
-
The simply supported beam is composed of two W12 \(\times 22\) sections built up as shown. Determine if the beam will safely support a loading of \(w=2 \mathrm{kip} / \mathrm{ft}\). The allowable...
-
If \(a=3 \mathrm{in}\). and the wood has an allowable normal stress of \(\sigma_{\text {allow }}=1.5 \mathrm{ksi}\), and an allowable shear stress of \(\tau_{\text {allow }}=150 \mathrm{psi}\),...
-
If \(P=800 \mathrm{lb}\), determine the minimum dimension \(a\) of the beam's cross section to the nearest \(\frac{1}{8}\) in. to safely support the load. The wood species has an allowable normal...

Study smarter with the SolutionInn App


