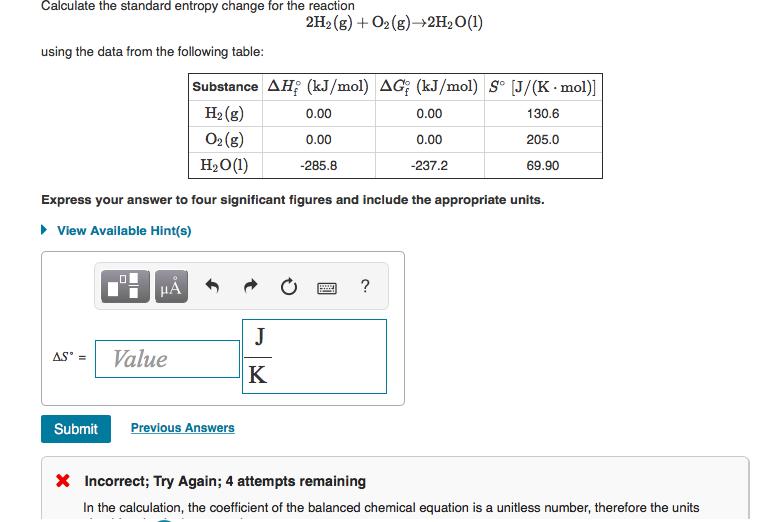
Calculate the standard entropy change for the reaction using the data from the following table: AS...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
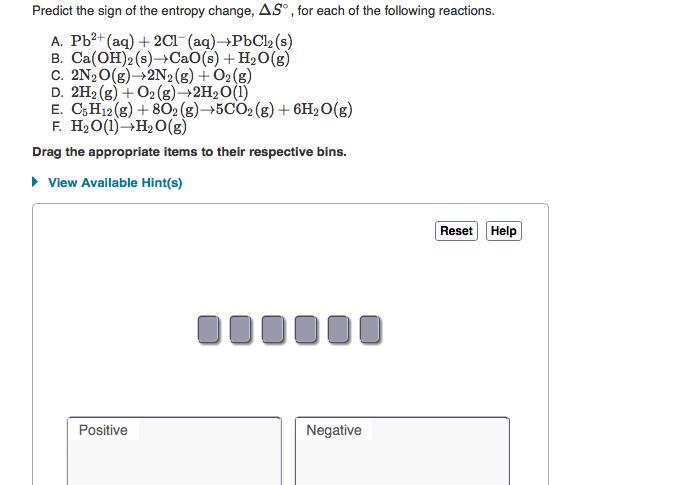
Calculate the standard entropy change for the reaction using the data from the following table: AS = HA Value Substance AH (kJ/mol) AG; (kJ/mol) So [J/(K mol)] H₂(g) 130.6 205.0 69.90 02 (8) H₂O (1) Express your answer to four significant figures and include the appropriate units. ▸ View Available Hint(s) 2H2(g) + O2(g) →→2H₂O(1) Submit Previous Answers J K 0.00 0.00 -285.8 0.00 0.00 -237.2 ? * Incorrect; Try Again; 4 attempts remaining In the calculation, the coefficient of the balanced chemical equation is a unitless number, therefore the units Predict the sign of the entropy change, AS, for each of the following reactions. A. Pb²+ (aq) + 2Cl(aq)→PbCl2 (s) B. Ca(OH)2 (s)→CaO(s) + H₂O(g) C. 2N2O(g) 2N2(g) + O2(g) D. 2H2(g) + O2(g) → 2H₂O(1) E. C5H12(g) + 802(g) →5CO2 (g) + 6H₂O(g) F. H₂O(1)→H₂O(g) Drag the appropriate items to their respective bins. ► View Available Hint(s) Positive 000000 Negative Reset Help Calculate the standard entropy change for the reaction using the data from the following table: AS = HA Value Substance AH (kJ/mol) AG; (kJ/mol) So [J/(K mol)] H₂(g) 130.6 205.0 69.90 02 (8) H₂O (1) Express your answer to four significant figures and include the appropriate units. ▸ View Available Hint(s) 2H2(g) + O2(g) →→2H₂O(1) Submit Previous Answers J K 0.00 0.00 -285.8 0.00 0.00 -237.2 ? * Incorrect; Try Again; 4 attempts remaining In the calculation, the coefficient of the balanced chemical equation is a unitless number, therefore the units Predict the sign of the entropy change, AS, for each of the following reactions. A. Pb²+ (aq) + 2Cl(aq)→PbCl2 (s) B. Ca(OH)2 (s)→CaO(s) + H₂O(g) C. 2N2O(g) 2N2(g) + O2(g) D. 2H2(g) + O2(g) → 2H₂O(1) E. C5H12(g) + 802(g) →5CO2 (g) + 6H₂O(g) F. H₂O(1)→H₂O(g) Drag the appropriate items to their respective bins. ► View Available Hint(s) Positive 000000 Negative Reset Help
Expert Answer:
Answer rating: 100% (QA)
A FB D Asswei f 24 g 29 20l D5 xxn 55 ss moduct as vendat 2x1306720503 Pb aq 2 aq Pbl... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Using the data in Appendix 3, calculate the standard entropy changes for the following reactions at 25C: (a) S(s) + O2(g) SO2(g) (b) MgCO3(s) MgO(s) + CO2(g)
-
Using the data in Appendix 3, calculate the standard entropy changes for the following reactions at 25C: (a) H2(g) + CuO(s) Cu(s) + H2O(g) (b) 2Al(s) + 3ZnO(s) Al2O3(s) + 3Zn(s) (c) CH4(g) + 2O2(g)...
-
Calculate the standard entropy change for the reaction at 25 C. Standard molar entropy values can be found in this. C,H,(g) + 50,(g) 3 CO,(g) + 4 H,0(g) ASixn J/K
-
Draw the Lewis structure of AlH 3 . Strategy Draw the Lewis structure in the normal way but realize that, in certain cases, it is not possible to place eight electrons around the central atom.
-
Is there a set career path in project management? Explain.
-
A polygon is a piecewise-linear, closed curve in the plane. That is, it is a curve ending on itself that is formed by a sequence of straight-line segments, called the sides of the polygon. A point...
-
Use the data in Short Exercise 16-12 to prepare Little People Learning Center's (LPLC) statement of cash flows for the year ended June 30, 2005. LPI.C uses the direct method for operating activities....
-
The PDC Company was described earlier in this chapter. Refer to the PDC Companys projected monthly operating schedules in Table. PDCs sales are projected to be $80,000 in September 2011. A. Prepare...
-
6.1 Audit evidence considerations (a) Discuss how analytical procedures can be used as substantive audit procedures to provide audit evidence. Illustrate your answer with an example. (b) (5 marks)...
-
You want to purchase a home based upon your current salary you decide that you can afford $2000.00 per month. Your bank has approved you for a (30 year) loan at an interest rate of 5%. 1) Based upon...
-
A person puts an empty chessboard on a table and puts one grain of rice on the first square. Then two grains on the second square, four on the third, eight on the fourth, and so forth, doubling the...
-
Someone says: Our firm focuses on maintaining long-term relationships with our customers. We dont have to do any prospecting. Evaluate this statement.
-
Give some examples of how CRM-enabled sales analytics can provide valuable insights at each of these stages of the customer journey: prospect (lead), initial customer contact, engagement with the...
-
Are there actually situations in which the use of one or more of the older sales technologies might be superior to something new? When and why would older approaches perhaps be a better choice in...
-
Consider any three of the newer types of sales technology presented in the chapter. For each, provide as many specific applications as you can think of that could enhance the buyerseller relationship...
-
Why do some salespeople not immediately embrace the use of the newest technologies? What are some ways in which a salesperson who is reluctant to adopt new technologies might be influenced to do so?
-
What does WHMIS stand for? 4 O Workplace Hazardous Management Information System O Workplace Hazardous Materials Information System. Workplace Hazardous Materials System
-
7 A 29-year-old, previously healthy man suddenly collapses at a party where legal and illicit drugs are being used. Enroute to the hospital, he requires resuscitation with defibrillation to establish...
-
Ksp for silver chloride at 25.0oC is 1.782 1010. At 35.0oC, Ksp is 4.159 1010. What are Ho and So for the reaction?
-
What is the age of a rock that contains equal numbers of 40 19 K and 40 18 Ar nuclei? The half-life of 40 19 K is 1.28 10 9 y.
-
What is the standard cell potential you would obtain from a cell at 25oC using an electrode in which Hg22+(aq) is in contact with mercury metal and an electrode in which an aluminum strip dips into a...
-
Jordan Quinn manages the local homeless shelter. He fears that a trusted employee has been stealing from the shelter. This employee receives cash from supporters and also prepares the monthly bank...
-
Refer to question 6. The balance of Allowance for Uncollectible Accounts, after adjustment, will be a. $ 100 $ 100 . b. $ 200 $ 200 . c. $ 300 $ 300 . d. impossible to determine from the information...
-
Refer to questions 6 and 7. Early the following year, Harper wrote off \(\$ 150\) of old receivables as uncollectible. The balance in the Allowance account is now The next four questions use the...

Study smarter with the SolutionInn App


