Element compound homogeneous mixture (heterogeneous mixture) 4) A piece of gold has a mass of 49.75...
Fantastic news! We've Found the answer you've been seeking!
Question:
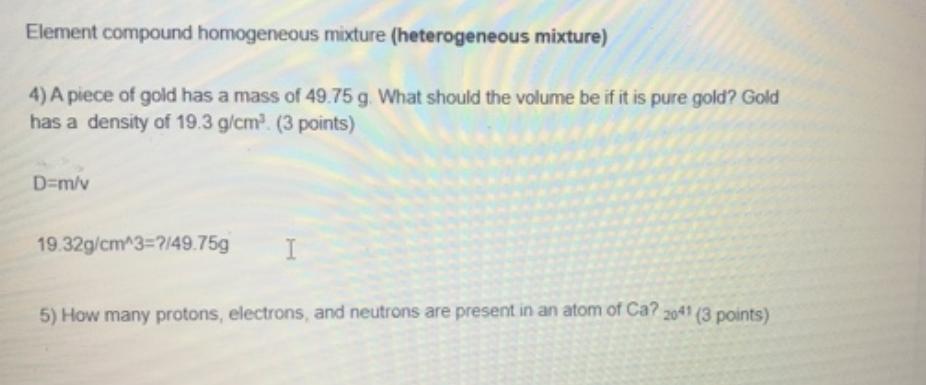
Transcribed Image Text:
Element compound homogeneous mixture (heterogeneous mixture) 4) A piece of gold has a mass of 49.75 g. What should the volume be if it is pure gold? Gold has a density of 19.3 g/cm³ (3 points) D=m/v 19.32g/cm^3=7/49.75g 5) How many protons, electrons, and neutrons are present in an atom of Ca? 2041 (3 points) Element compound homogeneous mixture (heterogeneous mixture) 4) A piece of gold has a mass of 49.75 g. What should the volume be if it is pure gold? Gold has a density of 19.3 g/cm³ (3 points) D=m/v 19.32g/cm^3=7/49.75g 5) How many protons, electrons, and neutrons are present in an atom of Ca? 2041 (3 points)
Expert Answer:
Answer rating: 100% (QA)
4 A piece of gold has a mass of 4975 g What should the volume be ... View the full answer

Related Book For 

Managerial Accounting
ISBN: 9780073526706
12th Edition
Authors: Ray H. Garrison, Eric W. Noreen, Peter C. Brewer
Posted Date:
Students also viewed these chemistry questions
-
A piece of gold has a mass of 20 g. If this gold were formed into a very thin sheet of thickness 0.50 mm, find the area of the sheet. Assume it has the same density as listed in Table 10.1. TABLE...
-
How high must a pointed arch be if it is to span a space 8.0m wide and exert one-third the horizontal force at its vase that a round arch would?
-
How long must a simple pendulum be if it is to make exactly one swing per second? (That is, one complete vibration takes exactly 2.0s)
-
Accurate Job Costing must be done on three levels. Which of the following is not one of these levels? Tracking and controlling costs during jobs Tracking gross profit each month Filing records on...
-
Richard and Charlotte can each choose only one of two locations to search for gold. The payoffs to each for each possible combination of choices are as given in the payoff matrix below. Which of the...
-
When you were looking at goodwill in the previous chapter, you were interested in the difference between the amount received and the value of net assets. Why do we consider only the assets when there...
-
What is the internal rate of return of the following cash flow diagram? a. 20 percent b. 18.2 percent c. 17.5 percent d. 15 percent $30 $31 0 1 2 3 $30 $15
-
American Auto is evaluating their marketing plan for the sedans, SUVs, and trucks they produce. A TV ad featuring this SUV has been developed. The company estimates that each showing of this...
-
in the COVID pandemic of 2020, the world's largest pharmaceutical companies were quick to develop vaccines. Would a market composed of many small companies have been able to respond as fast?
-
A chemical constituent flows between three reactors as depicted in Fig. P11.15. Steady-state mass balances can be written for a substance that reacts with first-order kinetics. For example, the mass...
-
Which do you prefer: a bank account that pays 5% per year (EAR) for three years or An account that pays 2.5% every six months for three years An account that pays 7.5% every 18 months for three years...
-
The CEO of Starbucks company wants to review the performance of the company in different countries during 2020. He knows that in 2020, the performance in all countries was affected by the COVID-19...
-
1. Explain the difference between hard and soft power. 2. Select 1 country from the list (see excel sheet in the Assignments folder), indicate on the sheet (no duplication), and identify with clear...
-
Mr.Mukund, who heads the production himself, developed an efficient top management group which includes Mr. Prem Heading marketing, Mrs. Mrunal leading HR and T&D, Mr.Vaidya leading Finance. All...
-
Choose the appropriate rule for the graph. Assume that dotted endpoints are included, and that undotted ones are not. Choose the appropriate rule for the graph below. OA. f(x) = { 2x + 2 - 1 2x+2 if...
-
The design for a full-adder shown in the figure below is used in the implementation of an 8-bit ripple carry adder in a processor ALU. The gates used in the full adder implementation have different...
-
An L 8 x 6 x 8 in. with staggered fasteners in each leg is shown. A36 steel is (Fy = 36 ksi, Fu = 50 ksi) and holes for 3/4 in. diameter bolts. a. Determine the design strength for LRFD b. Determine...
-
Problem 3.5 (4 points). We will prove, in steps, that rank (L) = rank(LT) for any LE Rnxm (a) Prove that rank (L) = rank (LTL). (Hint: use Problem 3.4.) (b) Use part (a) to deduce that that rank(L) =...
-
What adjustment is made for underapplied overhead on the schedule of cost of goods sold? What adjustment is made for overapplied overhead?
-
Doughboy Bakery would like to buy a new machine for putting icing and other toppings on pastries. These are now put on by hand. The machine that the bakery is considering costs $90,000 new. It would...
-
Define the following terms: differential cost, sunk cost, and opportunity cost.
-
Suppose we have a small inhabited island with three residents and a volcano that generates air pollution. Two people live upwind of the volcano and one person lives downwind. For \(\$ 21,000\) we can...
-
Assume a city of \(1,000,000\) people, \(60 \%\) of whom are willing to pay \(\$ 1\) maximum (each) to clean up pollution. The rest of the population is wealthier and is willing to pay \(\$ 100\)...
-
In Figure 3.2, using the Pareto criterion, which of the labeled points are socially preferred to \(W\) ? Figure 3.2 Brewster's utility B w' R Anna's utility S

Study smarter with the SolutionInn App


