Carefully study the following intramolecular Diels-Alder reaction scheme and answer the questions that follow. PLEASE NOTE:...
Fantastic news! We've Found the answer you've been seeking!
Question:
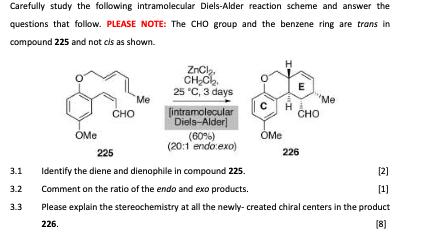
Transcribed Image Text:
Carefully study the following intramolecular Diels-Alder reaction scheme and answer the questions that follow. PLEASE NOTE: The CHO group and the benzene ring are trans in compound 225 and not cls as shown. 3.1 3.2 3.3 OMe CHO Me ZnCl₂ CH₂C₂₂ 25 °C, 3 days [intramolecular Diels-Alder] (60%) (20:1 endo:exo) с H OMe E CHO Me 225 Identify the diene and dienophile in compound 225. Comment on the ratio of the endo and exo products. Please explain the stereochemistry at all the newly-created chiral centers in the product 226. [8] 226 [2] [1] Carefully study the following intramolecular Diels-Alder reaction scheme and answer the questions that follow. PLEASE NOTE: The CHO group and the benzene ring are trans in compound 225 and not cls as shown. 3.1 3.2 3.3 OMe CHO Me ZnCl₂ CH₂C₂₂ 25 °C, 3 days [intramolecular Diels-Alder] (60%) (20:1 endo:exo) с H OMe E CHO Me 225 Identify the diene and dienophile in compound 225. Comment on the ratio of the endo and exo products. Please explain the stereochemistry at all the newly-created chiral centers in the product 226. [8] 226 [2] [1] Carefully study the following intramolecular Diels-Alder reaction scheme and answer the questions that follow. PLEASE NOTE: The CHO group and the benzene ring are trans in compound 225 and not cls as shown. 3.1 3.2 3.3 OMe CHO Me ZnCl₂ CH₂C₂₂ 25 °C, 3 days [intramolecular Diels-Alder] (60%) (20:1 endo:exo) с H OMe E CHO Me 225 Identify the diene and dienophile in compound 225. Comment on the ratio of the endo and exo products. Please explain the stereochemistry at all the newly-created chiral centers in the product 226. [8] 226 [2] [1] Carefully study the following intramolecular Diels-Alder reaction scheme and answer the questions that follow. PLEASE NOTE: The CHO group and the benzene ring are trans in compound 225 and not cls as shown. 3.1 3.2 3.3 OMe CHO Me ZnCl₂ CH₂C₂₂ 25 °C, 3 days [intramolecular Diels-Alder] (60%) (20:1 endo:exo) с H OMe E CHO Me 225 Identify the diene and dienophile in compound 225. Comment on the ratio of the endo and exo products. Please explain the stereochemistry at all the newly-created chiral centers in the product 226. [8] 226 [2] [1]
Expert Answer:
Answer rating: 100% (QA)
Answer Diene part Me Dienophile part ome 2 the dien... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Study the following case. Answer the questions that follow with Yes or No. Katsoulas, a U.S. car dealer, had a contract with a Korean automobile manufacturer for the delivery of several cars. When...
-
Study the following case. Answer the questions that follow with Yes or No. Straley Corporation, a manufacturer of hand tools, had been exporting its products to several countries in Europe for 10...
-
Study the following case. Answer the questions that follow with Yes or No. Bryerly, a U.S. sales representative for a U.S. firm, Zoom Airplane Company, was attempting to close a deal with a foreign...
-
An undamped mass spring system is released from rest from an initial displacement of x = 0.24 m and starts to oscillate. You see that the mass reaches its largest positive displacement for the first...
-
On January 1, 2011, Garner issued 10-year $200,000 face value, 6% bonds at par. Each $1,000 bond is convertible into 30 shares of Garner $2, par value, ordinary shares. Interest on the bonds is paid...
-
Is it typically necessary for a company to use an investment bank to conduct a merger? Mergers are fairly common in the U.S. There are a variety of reasons why mergers occur between two companies....
-
The breaking strength of hockey stick shafts made of two different graphite-kevlar composites yields the following results (in newtons). Can you conclude that the standard deviation of the breaking...
-
Planet Products, Inc., completed Job 2525 on May 31, and there were no jobs in process in the plant. Prior to June 1, the predetermined overhead application rate for June was computed from the...
-
George owns 100% of equity of a company. . If George works 6 hours per day, the company's EBIT will be 335,000 dollars per year, in perpetuity. If, instead, he works 12 hours per day, the firm's EBIT...
-
Zia Co. makes flowerpots from recycled plastic in two departments, Molding and Packaging. Zia uses the weighted average method, and units completed in the Molding department are transferred to the...
-
Historical trends over the last 3 years Calculate change in gross profit, operating profit & net profit from prior year. Calculate most current Gross Profit Margin, Operating Profit Margin & Net...
-
The following information has been provided by Miller Company: Advertising expense $ 2 0 , 6 0 0 Interest expense $ 8 , 2 0 0 Rent expense for store $ 2 5 , 6 0 0 Loss on sale of property and...
-
The Importance of Leadership Anyone who holds a responsible position in an organization must have a number of h qualities to meet his or her responsibilities successfully. One of the key qualities...
-
1) Leadership and management are very important vital components to an effective functioning organization so that people in the organization are inspired, managed and guided towards a common vision....
-
Assume you are the president of High Power Corporation. At the end of the first year of operations (December 31), the following financial data for the company are available: Accounts Payable $ 32,100...
-
Cupola Fan Corporation issued 1 0 % , $ 4 2 0 , 0 0 0 , 1 0 - year bonds for $ 4 0 3 , 0 0 0 on June 3 0 , 2 0 2 1 . Debt issue costs were $ 1 , 7 0 0 . Interest is paid semiannually on December 3 1...
-
ng and Technology consider a location where the wind with a density of 1.2 kg/m3 is blowing at a velocity of 4 m/s with a wind turbine with a rotor diameter of 1.03 m. find the maximum power that can...
-
Diamond Walker sells homemade knit scarves for $25 each at local craft shows. Her contribution margin ratio is 60%. Currently, the craft show entrance fees cost Diamond $1,500 per year. The craft...
-
Suggest how you would separate the free L-amino acid from its acylated D enantiomer in Figure. COOH COOH COOH H2N-CH CH-C NHC-H L is deacylated COOH O H-C-NH C CH L-amino acid CH C acylase COOH COOH...
-
Retroviruses like HIV, the pathogen responsible for AIDS, incorporate an RNA template that is copied into DNA during infection. The reverse transcriptase enzyme that copies RNA into DNA is relatively...
-
Triethyloxonium tetrafluoroborate, (CH3CH2)3O+ BF4-, is a solid with melting point 91-92 C. Show how this reagent can transfer an ethyl group to a nucleophile (Nuc:-) in an SN2 reaction. What is the...
-
Analytical procedures: a. Are intended to detect material misstatements in financial statement accounts. b. Are concerned with how internal control policies or procedures are applied. c. Are...
-
Substantive tests of details: a. Are intended to detect material misstatements in financial statement accounts. b. Are concerned with how internal control policies or procedures are applied. c. Are...
-
Tests of controls: a. Are intended to detect material misstatements in financial statement accounts. b. Are concerned with how internal control policies or procedures are applied. c. Are evaluations...

Study smarter with the SolutionInn App


