3. a. Acid catalyzed hydration of 2-methylpropene produces only 2-methyl-2- propanol. However, acid catalyzed of 1,2-dimethylcyclohexene...
Fantastic news! We've Found the answer you've been seeking!
Question:

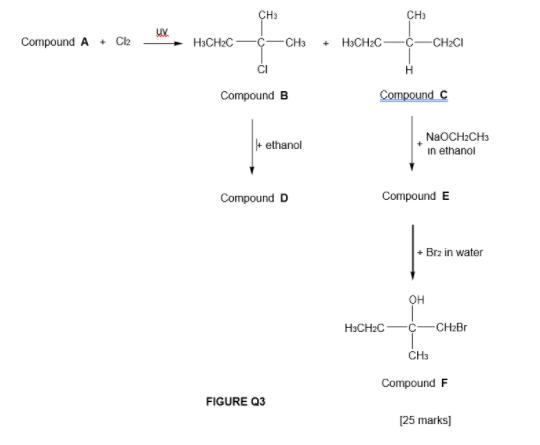
Transcribed Image Text:
3. a. Acid catalyzed hydration of 2-methylpropene produces only 2-methyl-2- propanol. However, acid catalyzed of 1,2-dimethylcyclohexene produces two products, ie, a mixture of stereoisomers. Using your knowledge in organic chemistry, discuss the regioselective and stereoselective nature of the acid catalyzed hydration based on reaction mechanism. [15 marks) b Compound A reacts with Clz in the presence of ultraviolet (UV) light to produce compounds B and C as shown in FIGURE Q3. Compound B reacts with ethanol at 60°C, to fom compound D (major product). Meanwhile compound C reacts with NaOCH,CH, in ethanol to form compound E. Compound E produces compound F when reacts with Br, in water. Using your knowledge in b. organic chemistry, explain in details the following. The constitutional isomers of compounds B and C, if any. The chirality of compounds B, C, D, E and F. If there islare enantiomer(s) among these compounds, show details of working steps in giving IUPAC name to the enantiomer(s). The difference in boiling point between compounds A, C and F. CH: CH) Compound A + C2 H3CH2C- CH3 + HƏCH:C- CH2CI CI Compound B Compound C NaOCH:CH3 + ethanol in ethanol Compound D Compound E + Brz in water он H>CH2C- CH2Br CH Compound F FIGURE Q3 [25 marks] 3. a. Acid catalyzed hydration of 2-methylpropene produces only 2-methyl-2- propanol. However, acid catalyzed of 1,2-dimethylcyclohexene produces two products, ie, a mixture of stereoisomers. Using your knowledge in organic chemistry, discuss the regioselective and stereoselective nature of the acid catalyzed hydration based on reaction mechanism. [15 marks) b Compound A reacts with Clz in the presence of ultraviolet (UV) light to produce compounds B and C as shown in FIGURE Q3. Compound B reacts with ethanol at 60°C, to fom compound D (major product). Meanwhile compound C reacts with NaOCH,CH, in ethanol to form compound E. Compound E produces compound F when reacts with Br, in water. Using your knowledge in b. organic chemistry, explain in details the following. The constitutional isomers of compounds B and C, if any. The chirality of compounds B, C, D, E and F. If there islare enantiomer(s) among these compounds, show details of working steps in giving IUPAC name to the enantiomer(s). The difference in boiling point between compounds A, C and F. CH: CH) Compound A + C2 H3CH2C- CH3 + HƏCH:C- CH2CI CI Compound B Compound C NaOCH:CH3 + ethanol in ethanol Compound D Compound E + Brz in water он H>CH2C- CH2Br CH Compound F FIGURE Q3 [25 marks]
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
When compound A is irradiated with ultraviolet light for 115 hours in pentane, an isomeric compound B is obtained that decolorizes bromine in CH2C12 and reacts with ozone to give, after the usual...
-
Explain why compound A reacts faster than compound B when they undergo solvolysis in aqueous acetone. CH C-Cl CH3 CH CH
-
When 2-methylpropene reacts with water and an acid catalyst, only one product alcohol is observed: tert-butyl alcohol (2-methyl-2-propanol). a. Draw the structures of the two intermediate...
-
IFRS requires the following: (a) Ending inventory is written up and down to net realizable value each reporting period. (b) Ending inventory is written down to net realizable value but cannot be...
-
A researcher wants to test the claim that the mean weight of male airline passengers, including carry-on baggage, is 195 lb. Identify the three different possible expressions that could be used for...
-
According to the National Association of Home Builders, the mean price of an existing single-family home in 2009 was $218,600. A real estate broker believes that because of the recent credit crunch,...
-
Why is the concept of DevOps appealing to organizations?
-
Turn to Part C of the Systems Analysts Toolkit and review the concept of net present value (NPV). Determine the NPV for the following: An information system will cost $95,000 to implement over a...
-
Lefrejun Ltd was formed nine months ago when Lee, Fred and June had finished their Bachelor of Construction Management programme at Deakin University. They saw an opportunity to sell wearable sensors...
-
Dorchester, Ltd. is an old-line confectioner specializing in high-quality chocolates. Through its facilities in the United Kingdom, Dorchester manufactures candies that it sells throughout Western...
-
Raw Materials Glucose 2. Cellular Respiration Products 1. Water 3. Answer the following questions in the spaces provided. 4. Where in the cell does the first stage of cellular respiration take place?...
-
Search and Rescue is the most important task assigned at the scene of an emergency. List and discuss at least 5 factors that a fire fighter must take into account when tasked with search and rescue...
-
Consider the following function over the given interval. f(x) = 16x2, [-4,4] b Evaluate the integral, f(x) dx. 256 3 Find the average value of the function over the given interval. (Round your answer...
-
Company purchases Deluxe machine for $52,000. It has an estimated salvage value of $2,000 and a useful life of four years. Company calculates the annual straight-line depreciation for the machine....
-
On 1 October 2020, Vanguard Ltd acquired 75% of Jupiter Ltd's equity shares by means of share exchange of two new shares in Vanguard Ltd for every five acquired shares in Jupiter. In addition,...
-
Using the values: locomotive1=steam locomotive2=diesel locomotive3=electric locomotive4=coal locomotive5=wood common=combined engines how do you write a Java program, using ArrayList, and input the...
-
Cannington Inc. designs, manufactures, and markets personal computers and related software. Cannington also manufactures and distributes music players (cPod), mobile phones (cPhone), and smartwatches...
-
Imagine you are the HR manager at a company, and a female employee came to you upset because she felt a male coworker was creating a hostile work environment by repeatedly asking her out on dates...
-
A national catalog and Internet retailer has three warehouses and three major distribution centers located around the country. Normally, items are shipped directly from the warehouses to the...
-
The traffic management office in Richmond is attempting to analyze the potential traffic flow from a new office complex under construction to an interstate highway interchange during the evening rush...
-
The bookstore at State University purchases from a vendor sweatshirts emblazoned with the school name and logo. The vendor sells the sweatshirts to the store for $38 apiece. The cost to the bookstore...
-
Describe the process of determining a nominal interest rate.
-
What risk factors should a team consider when deciding whether to build and fund a new venue? How are the risk factors different if a municipality is funding the construction?
-
Among NCAA mens basketball teams, which team would you expect to have the highest value? Why? How do you think conference affiliation affects value among these teams?

Study smarter with the SolutionInn App


