Deduce the structure of this compound using the spectroscopic information. Assign all significant peaks in all spectra.
Fantastic news! We've Found the answer you've been seeking!
Question:
Deduce the structure of this compound using the spectroscopic information. Assign all significant peaks in all spectra. 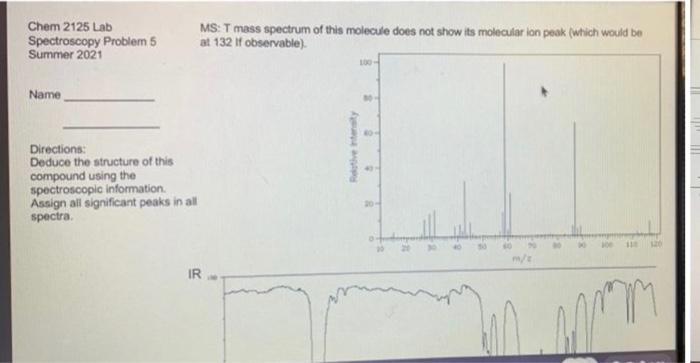
Transcribed Image Text:
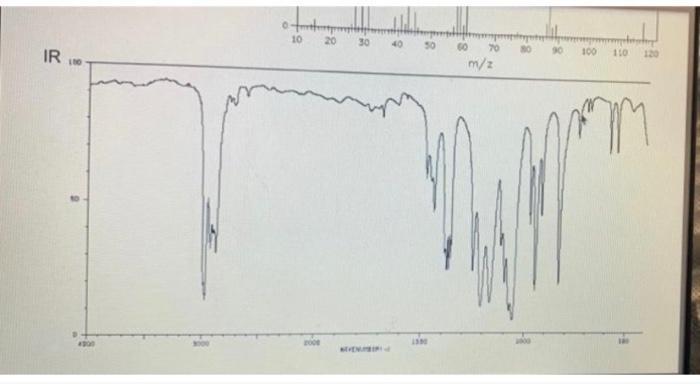
Chem 2125 Lab MS: T mass spectrum of this molecule does not show its molecular ion peak (which would be at 132 if observable). Spectroscopy Problem 5 Summer 2021 Name Directions: Deduce the structure of this compound using the spectroscopic information. Assign all significant peaks in all spectra. 20- m/e IR Retive intensity Chem 2125 Lab MS: T mass spectrum of this molecule does not show its molecular ion peak (which would be at 132 if observable). Spectroscopy Problem 5 Summer 2021 Name Directions: Deduce the structure of this compound using the spectroscopic information. Assign all significant peaks in all spectra. 20- m/e IR Retive intensity
Expert Answer:
Answer rating: 100% (QA)
From the IR spectra you can see a peak at around 1715 cm1 this peak corresponds to a carbony... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Deduce the structure of a compound having the mass spectrum and 1H NMR spectrum presented in Figure 13.43. Figure 13.43 100 80 E 60 e 40 20 134 10 20 30 40 50 60 70 80 90 100 110 120 30 140 50 nuz 15...
-
Deduce the structure of a compound with molecular formula C 9 H 10 O 2 that produces the following 1 H NMR spectrum and 13 C NMR spectrum: Proton NMR 10 Chemical Shift (ppm) Carbon NMR - 128.4 128.8-...
-
Deduce the structure of the compound that gives the following 1H, 13C, and IR spectra (Figs. 9.51-9.53). Assign all aspects of the 1H, and 13C spectra to the structure you propose. Use letters to...
-
A manufacturer of cases for sound equipment requires that holes be drilled for metal screws. The drill bits wear out and must be replaced; there is expense not only in the cost of the bits but also...
-
Dinnell Company owns the following assets: In the year of acquisition and retirement of an asset, Dinnell records depreciation expense for one- half year. During 2017, Asset A was sold for $ 7,000....
-
A machine component is subjected to the forces shown, each of which is parallel to one of the coordinate axes. Replace these forces with an equivalent force-couple system at A. imi 240N I50 N 125 30...
-
Refer to the Arctic Springs Bottling Department Data Set and. Complete Steps 3 and 4 of the process costing procedure: Summarize total costs to account for and then compute the cost per equivalent...
-
The following account titles were drawn from the general ledger of Holt Food Supplies, Incorporated (HFSI): Computers, Operating Expenses, Rent Revenue, Building, Cash, Notes Payable, Land, Utilities...
-
Automobile demand has fallen during a recent recession, and Ana has been laid off from her job on the assembly line. Rajiv recently lost his job as a waiter at a local restaurant. A recent increase...
-
On January 1, Year 4, Handy Company (Handy) purchased 70% of the outstanding common shares of Dandy Limited (Dandy) for $13,300. On that date, Dandy's shareholders' equity consisted of common shares...
-
Which of the following reactions is an endothermic reaction? i) Burning of coal ii) Decomposition of vegetable matter into compost iii) Process of respiration iv) Decomposition of calcium carbonate...
-
You are the payroll office of Wamco. You are new to the role. When reconciling the payroll reports, you noticed that employee Lisa Jones has had her superannuation guarantee underpaid for twelve...
-
1. A block of mass 2.0 kg passes at point A with a speed of 10 m/s. The length of |AB| is 2.0 m and |BC| is L. Coefficient of kinetic friction for the entire path is 0.2. What must L be so that the...
-
The market for stocks of small companies is less efficient than the market for stocks of large companies." Can you suggest a possible explanation for this observation?
-
Create an HTML and JavaScript documents to create and validate the following form: Personal Information Name (letters only-first letter capital): Date of Birth: Year (1960-2018): Month (01-12): | Day...
-
Sheffield Corporation had the following shareholders' equity on December 31, 2022: Common shares, 1,240,000 shares authorized, issued and outstanding Contributed surplus (Common Shares) Retained...
-
Explain how you went through the buyer decision-making process for your recent purchase of a high-involvement product (products that are normally expensive). Also mention how the...
-
Inexhaustible collections of ONPOs are not required to be capitalized or depreciated, if certain criteria are met. Why is this so, and what accounting and reporting recognition, if any, is required...
-
Show how you could prepare each of the following compounds from cyclopentanone, D2O, and any necessary organic or inorganic reagents. H OH
-
Write the structure of the enolate ion derived from each of the following -dicarbonyl compounds. Give the three most stable resonance forms of each enolate. (a) 1-Phenyl-1,3-butanedione (b) CH
-
The p-toluenesulfonate derived from (R)-2-octanol and ptoluenesulfonyl chloride was allowed to react with sodium benzenethiolate (C6H5SNa). Give the structure, including stereochemistry and the...
-
Which of the following best describes the underlying rationale for a written investment policy statement (IPS)? A. A written IPS communicates a plan for trying to achieve investment success. B. A...
-
Which of the following is least important as a reason for a written investment policy statement (IPS)? A. The IPS may be required by regulation. B. Having a written IPS is part of best practice for a...
-
A written investment policy statement (IPS) is most likely to succeed if: A. Created by a software program to assure consistent quality. B. It is a collaborative effort of the client and the...

Study smarter with the SolutionInn App


