Chemical reactions in foods that follow zero-order kinetics exhibit a constant rate of change in the...
Fantastic news! We've Found the answer you've been seeking!
Transcribed Image Text:
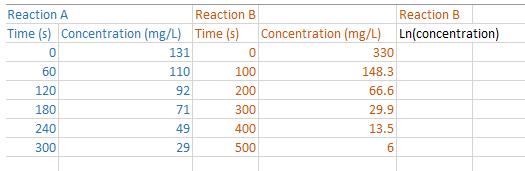
Chemical reactions in foods that follow zero-order kinetics exhibit a constant rate of change in the concentration of a reactant or a product. When the concentration is plotted against time, a straight line graph is obtained. The slope of the straight line gives the zero-order rate constant. On the other hand, many chemical reactions that occur during food processing and storage follow first-order kinetics. If we plot the concentration of a reactant against time we get an exponential type plot (i.e., a curve). The rate constant for a first- order reaction can be obtained by taking the natural logarithm of concentrations and then plotting those values against time. If the reaction follows first-order kinetics, then a plot of the natural logarithm of concentration against time should yield a straight line. Two sets of data are shown in the table below. a) Obtain a scatterplot of Concentration vs Time for Reaction A. (Be sure to put time on the x-axis). What trend do you see? Obtain a correlation coefficient. What does it tell you? What order of kinetics is being shown by Reaction A? b) Obtain a scatterplot of Concentration vs Time for Reaction B. (Be sure to put time on the x-axis). What trend do you see? What order of kinetics is being shown by Reaction B? c) Transform the concertation data for Reaction B by taking the natural logarithm. Obtain a scatterplot of Ln Concentration vs Time. What trend do you see now? d) Obtain a correlation coefficient for the relationship between Ln Concentration and Time. What does it tell you? Reaction A Reaction B Time (s) Concentration (mg/L) Time (s) 0 60 120 180 240 300 131 110 92 71 49 29 0 100 200 300 400 500 Concentration (mg/L) 330 148.3 66.6 29.9 13.5 6 Reaction B Ln(concentration) Chemical reactions in foods that follow zero-order kinetics exhibit a constant rate of change in the concentration of a reactant or a product. When the concentration is plotted against time, a straight line graph is obtained. The slope of the straight line gives the zero-order rate constant. On the other hand, many chemical reactions that occur during food processing and storage follow first-order kinetics. If we plot the concentration of a reactant against time we get an exponential type plot (i.e., a curve). The rate constant for a first- order reaction can be obtained by taking the natural logarithm of concentrations and then plotting those values against time. If the reaction follows first-order kinetics, then a plot of the natural logarithm of concentration against time should yield a straight line. Two sets of data are shown in the table below. a) Obtain a scatterplot of Concentration vs Time for Reaction A. (Be sure to put time on the x-axis). What trend do you see? Obtain a correlation coefficient. What does it tell you? What order of kinetics is being shown by Reaction A? b) Obtain a scatterplot of Concentration vs Time for Reaction B. (Be sure to put time on the x-axis). What trend do you see? What order of kinetics is being shown by Reaction B? c) Transform the concertation data for Reaction B by taking the natural logarithm. Obtain a scatterplot of Ln Concentration vs Time. What trend do you see now? d) Obtain a correlation coefficient for the relationship between Ln Concentration and Time. What does it tell you? Reaction A Reaction B Time (s) Concentration (mg/L) Time (s) 0 60 120 180 240 300 131 110 92 71 49 29 0 100 200 300 400 500 Concentration (mg/L) 330 148.3 66.6 29.9 13.5 6 Reaction B Ln(concentration)
Expert Answer:
Answer rating: 100% (QA)
A Obtain a scatterplot of Concentration vs Time for Reaction A Be sure to put time on the xaxis What trend do you see Obtain a correlation coefficient ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Doing calculations with relevant formulae (from lectures) and calculator is worth 8 marks, and spreadsheet work is worth 4 marks. For part (a), the spreadsheet work requires presenting this as an...
-
A car accelerates from rest at a constant rate of 2.0 m/s2 for 5.0 s. (a) What is the speed of the car at the end of that time? (b) How far does the car travel in this time?
-
A tire 0.500 m in radius rotates at a constant rate of 200 rev/min, find the speed and acceleration of a small stone lodged in the tread of the tire (on its outer edge).
-
Write the surface x 2 + y 2 z 2 = 2 (x + y) as an equation r = (, z) in cylindrical coordinates.
-
When would it be in a sellers interest to increase customer price-level awareness? When would it be in his or her interest to decrease customer price-level awareness?
-
Determine the values of c so that the following functions represent joint probability distributions of the random variables A" and Y: (a) f(x, y) cxy, for x = 1, 2, 3; y = 1, 2, 3; (b) f(x, y) = c\x...
-
Andrew Reitz established a trust in 2000, naming his sons, James and John, as sole beneficiaries and himself as trustee. Upon Andrews death, Hal Rachal Jr., the attorney who drafted the trust, became...
-
Rodman Corporations fiscal year ends on November 30. The following accounts are found in its job order cost accounting system for the first month of the new fiscal year. Other data: 1. On December 1,...
-
Objectives: Your company is currently planning to automate many different system administration tasks. Your manager has asked you to research a system administration task and implement it using...
-
7. Copper Mines, Inc (CMI) . purchased property for the purpose of extracting copper ore. CMl paid $3,500,000 for the property plus $100,000 closing costs. CMl estimates it will be able to extract...
-
Employees can leave the organisation for various reasons. Critically discuss the various reasons that employees would be dissatisfied in an organisation. In your discussion, include the different...
-
a. Explain how bar code readers work. b. Explain how optical character recognition (OCR) works and how it differs from bar code technology. c. Explain how scanners are used to capture data.
-
What is the purpose of the strategic IT plan?
-
Explain when a database designer might use a relationship table in constructing a relational database.
-
Explain the relationship between online real-time (OLRT) and immediate mode processing.
-
How does the COBIT framework define control?
-
Gleeson Ltd. has completed an initial public offering via a Dutch auction. Before the IPO, the firm had an existing capital of 1,200 million shares. The terms of the IPO were as follows: Initial...
-
Difference between truncate & delete
-
(a) For a parallel plate capacitor with a fixed plate separation distance, a larger plate area results in (1) a larger capacitance value, (2) an unchanged capacitance value, (3) a smaller capacitance...
-
Modern bowling alleys have automatic ball returns. The ball is lifted to a height of 2.00 m at the end of the alley and, starting from rest, rolls down a ramp. It continues to roll horizontally and...
-
A steam engine operates between 100 oC and 20 oC. What is the Carnot efficiency of the ideal engine that operates between these temperatures?
-
Deficits and fiscal consolidation As seen in the following table, the 2008 financial crisis left the United States with an enormous federal budget deficit in 2009. There was a substantial fiscal...
-
Which (if any) of the following statements about Pvalues is CORRECT? A. The Pvalue from a hypothesis test is the probability of obtaining your results, or more extreme results. B. The Pvalue from a...
-
Which (if any) of the following statements about the Type I error is CORRECT? A. The Type I error is the probability of rejecting the null hypothesis when it is true. B. The Type I error is the...

Study smarter with the SolutionInn App