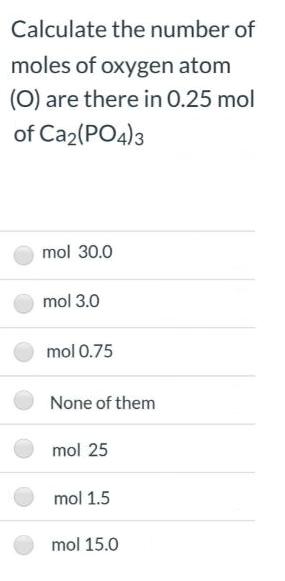
Calculate the number of moles of oxygen atom (0) are there in 0.25 mol of Ca2(PO4)3...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Calculate the number of moles of oxygen atom (0) are there in 0.25 mol of Ca2(PO4)3 mol 30.0 mol 3.0 mol 0.75 None of them mol 25 mol 1.5 mol 15.0 Calculate the number of moles of oxygen atom (0) are there in 0.25 mol of Ca2(PO4)3 mol 30.0 mol 3.0 mol 0.75 None of them mol 25 mol 1.5 mol 15.0
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the number of moles of HCl(g) that must be added to 1.0 L of 1.000 M NaC2H3O2 to produce a solution buffered at each pH. (Ka for HC2H3O2 = 1.800x10^-5) a) pH = pKa b) pH = 3.92 c) pH = 5.49
-
Calculate the number of moles of a gas that occupies a volume of 20 liters at a temperature of 25 C and 25 atm of pressure.n
-
Calculate the number of moles of barium chloride in 427 g of a 3.17% by mass barium chloride solution? a. 6.50 10 02 mol b. 7.83 10 02 mol c. 4.31 10 02 mol d. 7.81 10 02 mol e. 4.27 10 02 mol
-
Suppose the following game is repeated once (that is, played twice in total). Bridget Alex SB (40,40) (70,20) SA SA (20, 70) (30,30) (a) How many strategies does Alex have? (b) In an SPNE, what...
-
If, based on a sample size of 850, a political candidate finds that 458 people would vote for him in a two person race, what is the 95% confidence interval for his expected proportion of the vote?...
-
The Quinnipiac University Poll conducts nationwide surveys as a public service and for research. This problem is based on the results of one such poll taken in May 2008. Independent simple random...
-
For approximately 20 months, Robert E. McDonald perpetrated a scheme to solicit millions of dollars purportedly for a \($100\) million purchase by the RAI Entities and certain other related corporate...
-
American Products is concerned about managing cash efficiently. On the average, inventories have an age of 90 days, and accounts receivable are collected in 60 days. Accounts payable are paid...
-
Consider two oppositely charged atoms with charges of +1 and -1 units, respectively. The two atoms interact with each other through electrostatic (Coulombic) and van der Waals forces, and reach an...
-
An example in this chapter referred to a survey of 410 human resource professionals. If those subjects are identified with numbers from 1 through 410, does it make sense to calculate the average...
-
Which of the following is an example of quantitative data? Oorder in which students complete a test. social security numbers O telephone numbers O number of children in a family
-
Describe how an auditor determines their audit strategy and relationship between preliminary risk assessments and audit strategy. Explain and appraise 4 different types of audit evidence and...
-
JoJo Company signed a 18-month, non-interest-bearing note on October 31 for the purchase of $150,000 of merchandise. The face value of the note was $163,230. JoJo elects to use a discount account for...
-
Klein Corporation's statement of cash flows showed the following activities for the year ended December 31, 2022. Cash from operating activities: $ 30,000 Cash from investing activities: 15,000 Cash...
-
A. Evaluate the effectiveness of your goal and project outcome by doing the following: 1. Compare the results of two key performance indicators (KPIs) to the SMART+C goal and outcome statement you...
-
Answer the following questions after reading the article Below Discuss some of the similarities between the coast guard and accountants; do you agree with the similarities? Why or why not? What do...
-
A simple camera telephoto lens consists of two lenses. The objective lens has a focal length f = +42.0 cm. Precisely 34.8 cm behind this lens is a concave lens with a focal length f = -13.8 cm. The...
-
Distinguish between the work performed by public accountants and the work performed by accountants in commerce and industry and in not-for-profit organisations.
-
Sodium-23 is the only stable isotope of sodium. Predict how sodium-20 will decay and how sodium-26 will decay.
-
Give the normal state (solid, liquid, or gas) of each of the following. a. Sodium hydrogen carbonate (baking soda) b. Isopropyl alcohol (rubbing alcohol) c. Oxygen d. Copper
-
An aqueous solution of an unknown salt of vanadium is electrolyzed by a current of 2.50 amps for 1.90 hours. The electroplating is carried out with an efficiency of 95.0%, resulting in a deposit of...
-
Identify and correct the errors in the following passages. a. Company's are finding it to their advantage to cultivate their suppliers. Partnerships among a company and it's suppliers can yield hefty...
-
How can you create an effective strategy for your interview?
-
How can you plan for a successful career?

Study smarter with the SolutionInn App



