Question: 6. Complete the following reactions by filling what is left out; either the reactants, starting material, or the major product. (5 pts ea) a.
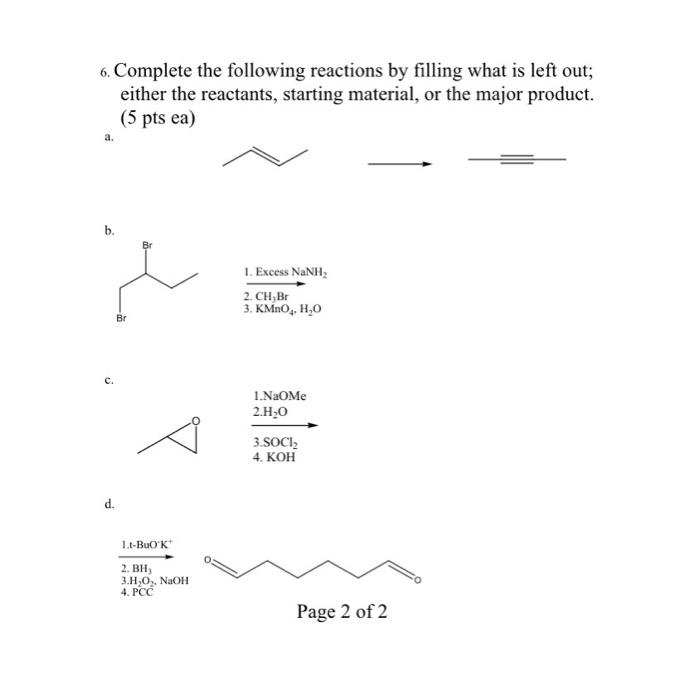
6. Complete the following reactions by filling what is left out; either the reactants, starting material, or the major product. (5 pts ea) a. b. d. Br 1.t-BuOK 2. BH, 3.HO, NaOH 4. PCC 1. Excess NaNH, 2. CH,Br 3. KMnO4, HO 1.NaOMe 2.HO 3.SOCI 4. KOH Page 2 of 2
Step by Step Solution
3.51 Rating (175 Votes )
There are 3 Steps involved in it
A Bromination followed by dehydrohalogenation B Dehydrohalogenation followed b... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
635d91e5b77fe_176848.pdf
180 KBs PDF File
635d91e5b77fe_176848.docx
120 KBs Word File


