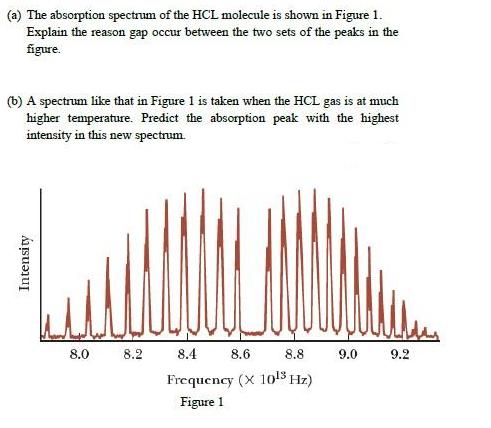
(a) The absorption spectrum of the HCL molecule is shown in Figure 1. Explain the reason...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
(a) The absorption spectrum of the HCL molecule is shown in Figure 1. Explain the reason gap occur between the two sets of the peaks in the figure. (b) A spectrum like that in Figure 1 is taken when the HCL gas is at much higher temperature. Predict the absorption peak with the highest intensity in this new spectrum. 8.0 8.2 8.4 8.6 8.8 9.0 9.2 Frequency (X 1o!5 Hz) Figure 1 Intensity (a) The absorption spectrum of the HCL molecule is shown in Figure 1. Explain the reason gap occur between the two sets of the peaks in the figure. (b) A spectrum like that in Figure 1 is taken when the HCL gas is at much higher temperature. Predict the absorption peak with the highest intensity in this new spectrum. 8.0 8.2 8.4 8.6 8.8 9.0 9.2 Frequency (X 1o!5 Hz) Figure 1 Intensity
Expert Answer:
Answer rating: 100% (QA)
1 A classic among molecular spectra the infrared absorption spectrum of HCl can be analyzed to gain information about both rotation and vibration of the molecule The absorption lines shown involve tra... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A system with unity feedback is shown in Figure E5.11. Determine the steady-state error for a step and a ramp input when Figure E5.11 Unity feedback system. 5(s + 8) s(s 1)(s 4)(s 10) G(s) Gs) Yts)
-
A spacecraft with a camera is shown in Figure AP6.6(a).The camera slews about 16° in a canted plane relative to the base. Reaction jets stabilize the base against the reaction torques from the...
-
A cooling curve is shown in Figure 9 - 21. Determine (a) The pouring temperature; (b) The solidification temperature; (c) The superheat; (d) The cooling rate, just before solidification begins; (e)...
-
HL Co. uses the high-low method to derive a total cost formula. Using a range of units produced from 1,500 to 7,500, and a range of total costs from $21,000 to $45,000, producing 2,000 units will...
-
The New York Attorney General began an investigation of Monster Energy Drinks, Pepsis AMP, and 5-Hour Energy Drinks to determine whether the companies are adequately disclosing the amount of caffeine...
-
Sherry, who is 52 years of age, opened a Roth IRA three years ago. She has contributed a total of $12,000 to a Roth IRA ($4,000 a year).The current value of the Roth IRA is $16,300.In the current...
-
There is a current of \(0.25 \mathrm{~A}\) in the circuit of Figure P23.65. a. What is the direction of the current? Explain. b. What is the value of the resistance \(R\) ? c. What is the power...
-
Kroger Co.??s 2009 financial statements contained the following data (in millions). InstructionsCompute these values:(a) Working capital. (b) Currentratio. Current assets Total assets Current...
-
Suppose that, for 2010, properties in Paragon are not reassessed but that new construction increases total assessed value by 9 percent. The state enacts a new property tax control that allows the...
-
It is October 16, 2020, and you have just taken over the accounting work of China Moon Products, whose annual accounting period ends October 31. The company?s previous accountant journalized its...
-
Exercise 4.28 Let a and a1,..., am be given vectors in R". Prove that the following two statements are equivalent: (a) For all x > 0, we have a'x < max, ax. (b) There exist nonnegative coefficients...
-
Blair sells an acre of land (with potential environmental issues) to Abby for $200,000. Blair receives a demand promissory note with a face amount of $200,000 bearing adequate interest. However, due...
-
2. Firms HD and LD are identical except for their use of debt and the interest rates they pay--HD has more debt and thus must pay a higher interest rate. Based on the data given below, how much...
-
One year ago today you bought a 6% coupon bond with a face value of 100 and 5 years to maturity for a price of 102.47. The bond pays interest annually. In the meantime the yield to maturity for the...
-
Beyonce went to the mall and saw a massage chair that she would have to take a loan out for $6,500 to purchase. The bank said that she could get a simple interest rate of 8% for 5 years. What is the...
-
When a 500-g statue of a falcon suspended from a spring is immersed in water, the scale reads 400 g, and when immersed in benzene, the scale reads 412 g. Determine the density of benzene.
-
You have a choice between two construction sites on a natural slope, both underlain by a weak shale. At one site, the beds dip into the hill (fig. a); at the other, the beds dip parallel to the slope...
-
At the beginning of its fiscal year, Lakeside Inc. leased office space to LTT Corporation under a seven-year operating lease agreement. The contract calls for quarterly rent payments of $25,000 each....
-
An adaptive suspension vehicle uses a legged locomotion principle. The control of the leg can be represented by a unity feedback system with [12] We desire to achieve a steady-state error for a ramp...
-
A system has the transfer function (a) Construct a state variable representation of the system. (b) Determine the element Ï11(t) of the state transition matrix for this system. Y(s) R(s)
-
One of the most potentially beneficial applications of an automotive control system is the active control of the suspension system. One feedback control system uses a shock absorber consisting of a...
-
Gavins Gardening Equipment Hires unadjusted trial balance of the business appears as shown below. Ignore GST. Additional information 1. Expired insurance amounts to $750. 2. June electricity costs of...
-
Lana Priest set up a home sewing business on 1 July 2019. Usually, Lana collects $20 per hour for sewing on the completion of each days work and pays for the maintenance of her machine with cash....
-
Craigs Car Detailing Service had the following accounts and account balances in the adjusted trial balance columns of its worksheet for the year ended 30 June 2019. Required (a) Record the required...

Study smarter with the SolutionInn App


