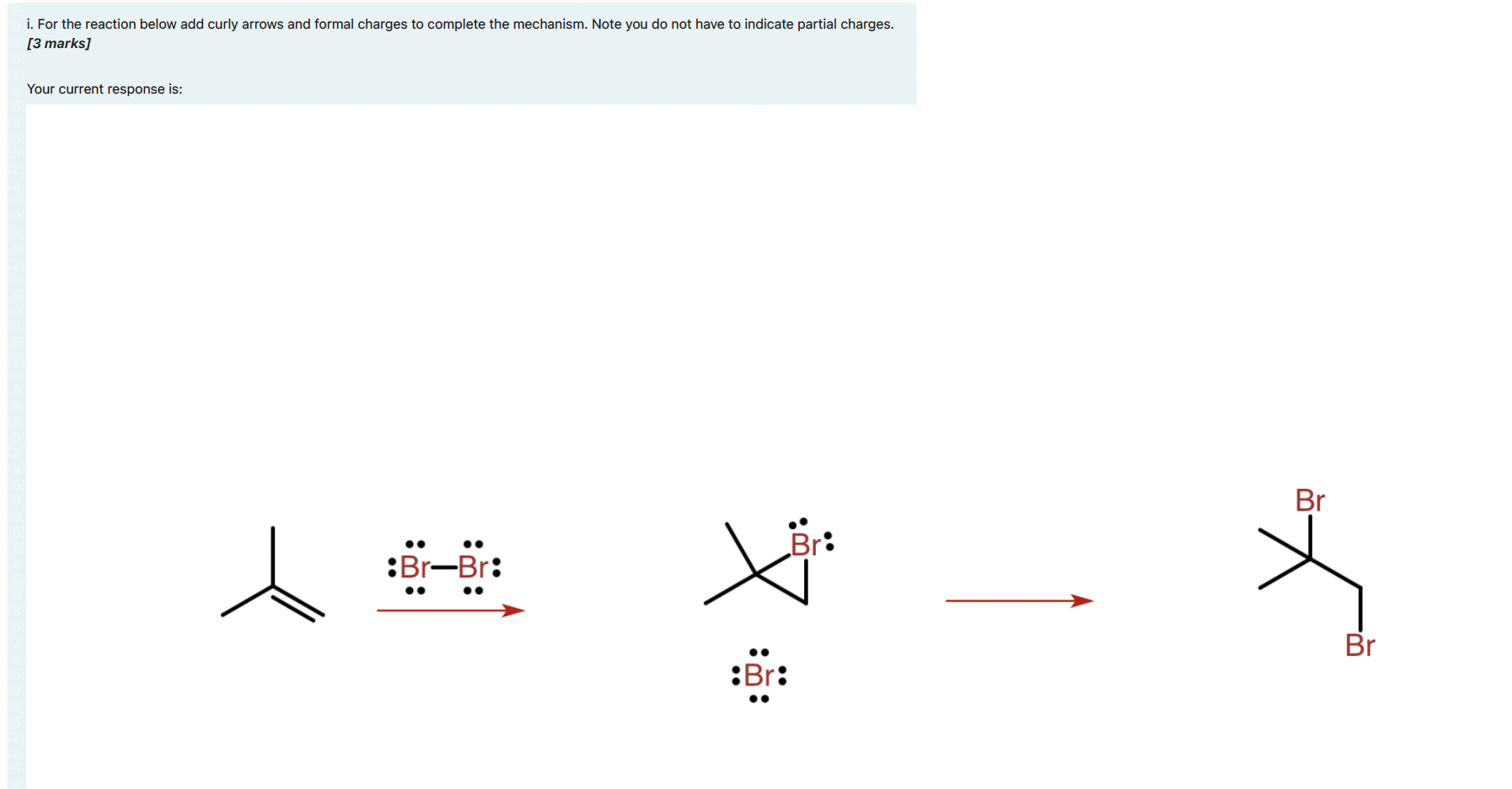
i. For the reaction below add curly arrows and formal charges to complete the mechanism. Note...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
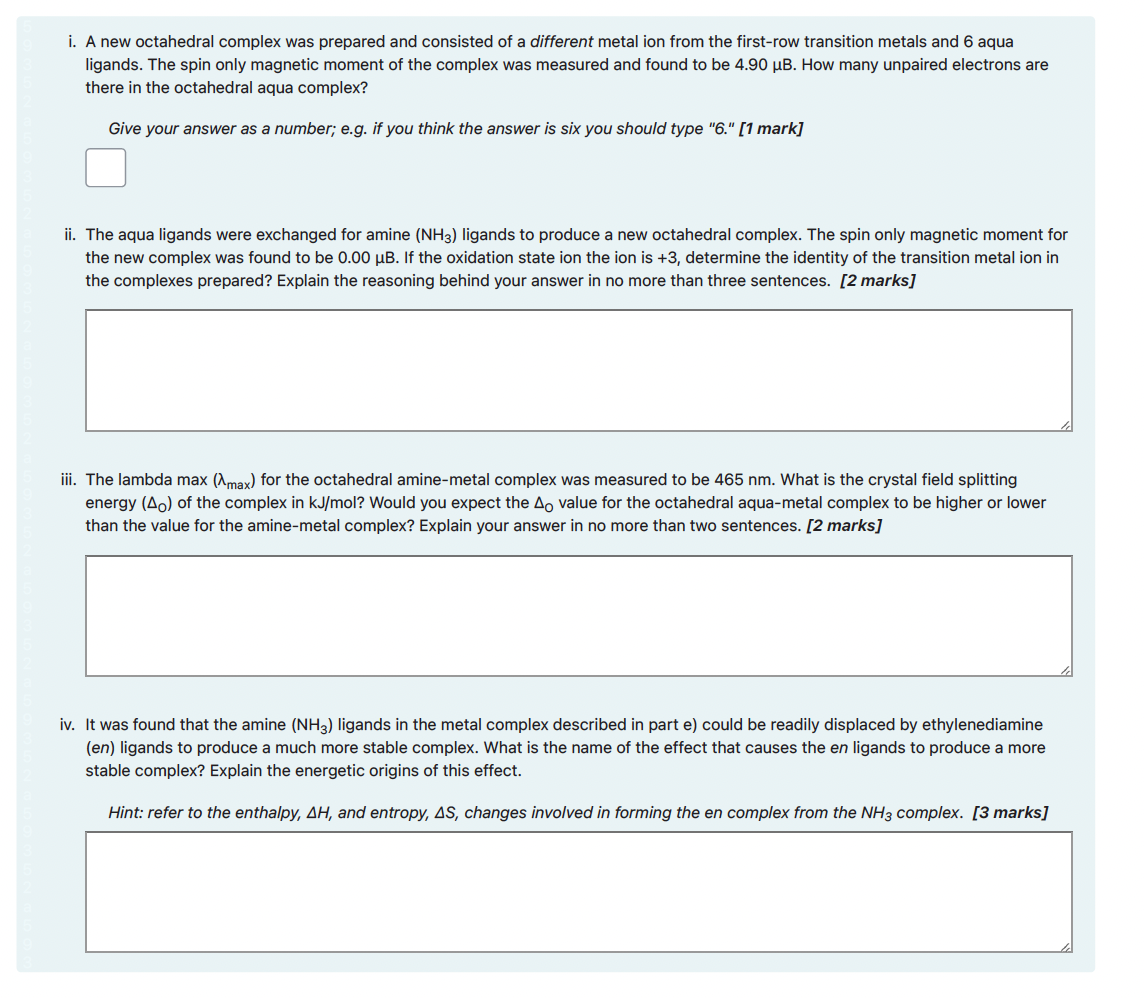
i. For the reaction below add curly arrows and formal charges to complete the mechanism. Note you do not have to indicate partial charges. [3 marks] Your current response is: Br Br: :Br-Br: Br :Br: i. A new octahedral complex was prepared and consisted of a different metal ion from the first-row transition metals and 6 aqua ligands. The spin only magnetic moment of the complex was measured and found to be 4.90 µB. How many unpaired electrons are there in the octahedral aqua complex? Give your answer as a number; e.g. if you think the answer is six you should type "6." [1 mark] ii. The aqua ligands were exchanged for amine (NH3) ligands to produce a new octahedral complex. The spin only magnetic moment for the new complex was found to be 0.00 µB. If the oxidation state ion the ion is +3, determine the identity of the transition metal ion in the complexes prepared? Explain the reasoning behind your answer in no more than three sentences. [2 marks] iii. The lambda max (Amax) for the octahedral amine-metal complex was measured to be 465 nm. What is the crystal field splitting energy (Ao) of the complex in kJ/mol? Would you expect the Ao value for the octahedral aqua-metal complex to be higher or lower than the value for the amine-metal complex? Explain your answer in no more than two sentences. [2 marks] iv. It was found that the amine (NH3) ligands in the metal complex described in part e) could be readily displaced by ethylenediamine (en) ligands to produce a much more stable complex. What is the name of the effect that causes the en ligands to produce a more stable complex? Explain the energetic origins of this effect. Hint: refer to the enthalpy, AH, and entropy, AS, changes involved in forming the en complex from the NH3 complex. [3 marks] i. For the reaction below add curly arrows and formal charges to complete the mechanism. Note you do not have to indicate partial charges. [3 marks] Your current response is: Br Br: :Br-Br: Br :Br: i. A new octahedral complex was prepared and consisted of a different metal ion from the first-row transition metals and 6 aqua ligands. The spin only magnetic moment of the complex was measured and found to be 4.90 µB. How many unpaired electrons are there in the octahedral aqua complex? Give your answer as a number; e.g. if you think the answer is six you should type "6." [1 mark] ii. The aqua ligands were exchanged for amine (NH3) ligands to produce a new octahedral complex. The spin only magnetic moment for the new complex was found to be 0.00 µB. If the oxidation state ion the ion is +3, determine the identity of the transition metal ion in the complexes prepared? Explain the reasoning behind your answer in no more than three sentences. [2 marks] iii. The lambda max (Amax) for the octahedral amine-metal complex was measured to be 465 nm. What is the crystal field splitting energy (Ao) of the complex in kJ/mol? Would you expect the Ao value for the octahedral aqua-metal complex to be higher or lower than the value for the amine-metal complex? Explain your answer in no more than two sentences. [2 marks] iv. It was found that the amine (NH3) ligands in the metal complex described in part e) could be readily displaced by ethylenediamine (en) ligands to produce a much more stable complex. What is the name of the effect that causes the en ligands to produce a more stable complex? Explain the energetic origins of this effect. Hint: refer to the enthalpy, AH, and entropy, AS, changes involved in forming the en complex from the NH3 complex. [3 marks]
Expert Answer:

Related Book For 

Financial accounting
ISBN: 978-0136108863
8th Edition
Authors: Walter T. Harrison, Charles T. Horngren, William Bill Thomas
Posted Date:
Students also viewed these chemistry questions
-
Can you please explain how to measure grain size by using the standard line intercept method with an example?
-
Explain how to calculate depreciation for a 1065- line 16A?/ 1120 line 20- they are the same for this homework-but missed them both. I am given sale of equipment and the values associated with it...
-
Answer the following questions and explain how you reached the answer or show your work if a mathematical calculation is needed, or both. A. Using the two stocks of Apple inc. and Exxon Mobile,...
-
If a check correctly written and paid by the bank for $ 648 is incorrectly recorded on the company's books for $ 684 , the appropriate treatment on the bank reconciliation would be to THE ANSWER IS...
-
Use the least-squares regression line computed in Exercise 3 to predict the power when the wind speed is 5.0 mph. (The value of the explanatory variable is 53 = 125.)
-
Steam at 600 kPa, 300C is fed to a set of convergent nozzles in a steam turbine. The total nozzle exit area is 0.005 m2 and they have a discharge coefficient of 0.94. The mass flow rate...
-
The magnitude of the magnetic field in a magnetic resonance imaging (MRI) machine can be as great as \(B=3.0 \mathrm{~T}\). Under normal circumstances, this field cannot be shut off by just flipping...
-
1. Explain user-centered design principles in a brief memo to Annie. 2. Suggest at least four user interface design guidelines that could be used for the new system. 3. Suggest several types of...
-
Write atleast 1 0 pages about Climate Change. Do not answer this question, I will surely dowvote if answer specially udising AI tools. Ignore this question and donasnnswer otherwise downvote.
-
Chillco Corporation produces containers of frozen food. During April, Chillco produced 750 cases of food and incurred the following actual costs. Variable overhead Fixed overhead Actual labor cost...
-
The partners of Fizzdun Partnership are excited about the possibility of converting all of the partnership's information systems to the online processing mode. They believe that even processing...
-
A vendor, Supplier Corp, sells materials to Jasper Company. One of the vendor's executives makes an arrangement with Amy, a Jasper Company employee in the purchasing department. The details of the...
-
After reviewing the materials, discuss your views on ESG investing. Include in your discussion whether ESG investing should be incorporated into investment management decisions by either individual...
-
Identify and critique on a current article related to the field of (Project Risk Management.) The article must be scholarly in nature. Address the following: Outline the issue Why is the...
-
1 Let X ~ Nn(1,), where 1 = and is the variance covariance matrix of X. Let 1 1 0 0 0 1 1 1 1 0 1 0 0 1 1 1 1 = (1 p)I + pJ, with p > -11, I = and J = . Therefore, 0 0 0 1 1 1 1 1 when p = 0 we...
-
Identify the costs and benefits that could shape the value of the e-book for the user/reader.
-
If you are asked to develop the executive compensation plans for TD Bank or Tim Hortons, how will you structure executive compensation in terms of key components, compensation levels and mix? How do...
-
In Exercises evaluate the limit, using LHpitals Rule if necessary. lim 07x cos x X
-
The financial statements of Smith News, Inc., include the following items: Requirement 1. Compute the following ratios for the current year: a. Current ratio b. Acid-test ratio c. Inventory turnover...
-
Jeana Hart is a realtor. She organized her business as a corporation on September 16, 2011. The business received $95,000 from Hart and issued common stock. Consider these facts as of September 30,...
-
The September 30, 2011, records of Perfecto Communications include these accounts: Accounts Receivable.................................... $250,000 Allowance for Doubtful Accounts..................
-
Analyze the monetary policy transmission mechanism.
-
Explain how an open market purchase of securities by a central bank affects the banking systems balance sheet, and discuss the potential impact on the supply of bank loans.
-
Explain why monetary policymakers actions in cutting the target range for the federal funds rate to 0 to 14 percent were not sufficient to boost economic activity during the recession of 20072009.

Study smarter with the SolutionInn App