a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest...
Fantastic news! We've Found the answer you've been seeking!
Question:
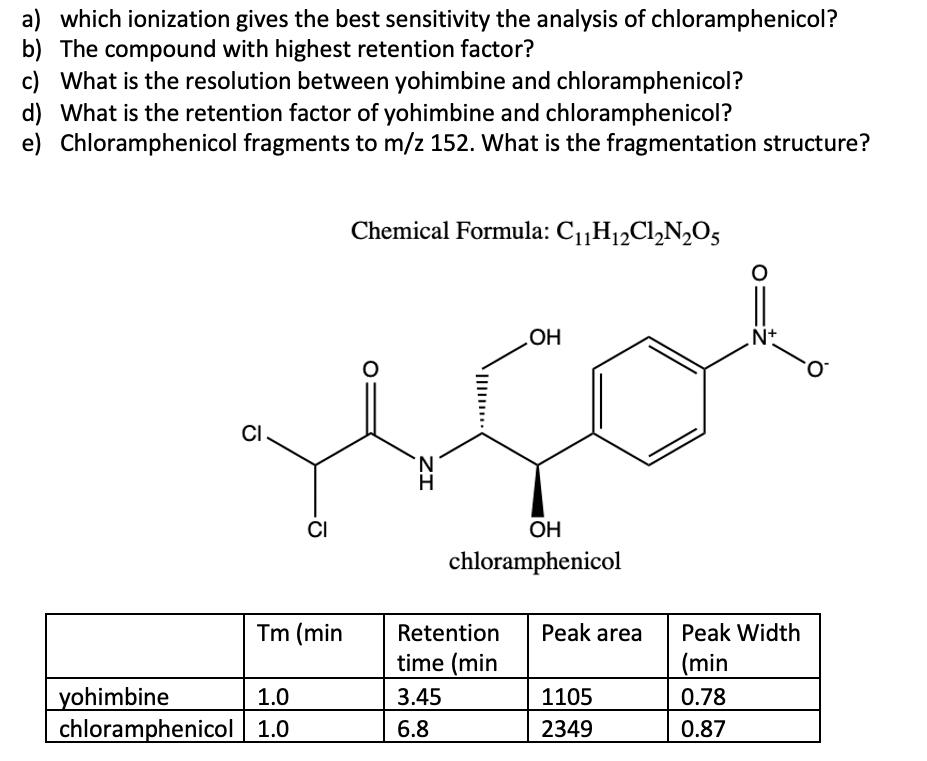
Transcribed Image Text:
a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest retention factor? c) What is the resolution between yohimbine and chloramphenicol? d) What is the retention factor of yohimbine and chloramphenicol? e) Chloramphenicol fragments to m/z 152. What is the fragmentation structure? Chemical Formula: C1„H12C1,N,O5 HO Nt O. CI. CI OH chloramphenicol Tm (min Retention Peak area Peak Width time (min (min yohimbine chloramphenicol 1.0 1.0 3.45 1105 0.78 6.8 2349 0.87 ZI a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest retention factor? c) What is the resolution between yohimbine and chloramphenicol? d) What is the retention factor of yohimbine and chloramphenicol? e) Chloramphenicol fragments to m/z 152. What is the fragmentation structure? Chemical Formula: C1„H12C1,N,O5 HO Nt O. CI. CI OH chloramphenicol Tm (min Retention Peak area Peak Width time (min (min yohimbine chloramphenicol 1.0 1.0 3.45 1105 0.78 6.8 2349 0.87 ZI
Expert Answer:
Answer rating: 100% (QA)
a The electrospray negative ionization Electrospray Ionization ESI probe in negative ionization mode ... View the full answer

Related Book For 

Applied Statistics in Business and Economics
ISBN: 978-0073521480
4th edition
Authors: David Doane, Lori Seward
Posted Date:
Students also viewed these chemistry questions
-
Compound C (C9H11NO) gives a positive Tollens' test (can be oxidized to a carboxylic acid) and is soluble in dilute HCl. The IR spectrum of C shows a strong band near 1695 cm-1 but shows no bands in...
-
A compound with molecular formula C4H6O gives the infrared spectrum shown in Figure 13.34. Identify the compound. 3.5 4 5 12 13
-
compound with molecular formula C5H10O2 gives the following IR spectrum. When it undergoes acid-catalyzed hydrolysis, the compound with the following 1H NMR spectrum is formed. Identify the...
-
Prove the following statement: If a transformer having a series impedance Ze is connected as an autotransformer, its per-unit series impedance Z as an autotransformer will be SE Zoq NSE + NC Note...
-
A length of wire is cut in half and the two lengths are wrapped together side by side to make a thicker wire. How does the resistance of this new combination compare to the resistance of the original...
-
The number of hits to a website follows a Poisson process and occurs at the rate of 10 hits per minute between 7:00 p.m. and 9:00 p.m. How long should you expect to wait before the probability of at...
-
At a relatively close 51.4 kiloparsecs away from the Earth, the supernova of 1987 was the closest nova to occur since telescopes were invented, and it allowed a unique opportunity to confirm that...
-
The Eatery is a restaurant in DeKalb, Illinois. It specializes in deluxe sandwiches in a moderate price range. Michael Raye, the manager of The Eatery, has determined that during the last 2 years the...
-
True or False: Excels NPV function treats CF in year 1 as the CF in year 2.
-
Identify the process evaluation article that you chose and explain why you selected this example. Describe the purpose of the evaluation, the informants, the questions asked, and the results of the...
-
Question 25 25) Evaluate the following expression exactly O .3141593 using trigonometric identities. O .1766413 sin 10 + cost O .9945319 10 O
-
Pacifico Company, a U . S . - based importer of beer and wine, purchased 1 , 7 0 0 cases of Oktoberfest - style beer from a German supplier for 4 5 9 , 0 0 0 euros. Relevant U . S . dollar exchange...
-
Consider each of the following scenarios and identify a behavioral intervention to address each issue in family work. A teenager not complying with curfew. One member of the couple not picking up...
-
Sandy Crane Hospital expanded its maternity ward to add patient rooms for extended hospital stays. They negotiated a 15-year loan with monthly payments and a large sum of $250,000 due at the end of...
-
2 (39 marks) R QUESTION 2 (39 marks) Roundworm Ltd is a group of companies with a 31 December year-end. The Roundworm group financial statements for the years 20.21 and 20.22 are given below:...
-
Vino Veritas Company, a U.S.-based importer of wines and spirits, placed an order with a French supplier for 1,400 cases of wine at a price of 240 euros per case. The total purchase price is 336,000...
-
The following information relates to Franklin Freightways for its first year of operations (data in millions of dollars): Pretax accounting income: $ 767 Pretax accounting income included: Overweight...
-
Consider the activities undertaken by a medical clinic in your area. Required 1. Do you consider a job order cost accounting system appropriate for the clinic? 2. Identify as many factors as possible...
-
Engineers are testing company fleet vehicle fuel economy (miles per gallon) performance by using different types of fuel. One vehicle of each size is tested. Does this sample provide sufficient...
-
At a University of Colorado woman's home basketball game, a random sample of 25 con- cession purchases showed a mean of $7.12 with a standard deviation of $2.14. For the next week's home game, the...
-
A manufacturing project has five independent phases whose completion must be sequential. The time to complete each phase is a random variable. The mean and standard deviation of the time for each...
-
Refer to the completely randomized design of Example 10.4, in which we concluded that at least two of the four brands of golf balls are associated with different mean distances traveled when struck...
-
Chemical properties of whole wheat breads. Whole wheat breads contain a high amount of phytic acid, which tends to lower the absorption of nutrient minerals. The Journal of Agricultural and Food...
-
A multiple-comparison procedure for comparing four treatment means produced the confidence intervals shown here. Rank the means from smallest to largest. Which means are significantly different? LO4...

Study smarter with the SolutionInn App


