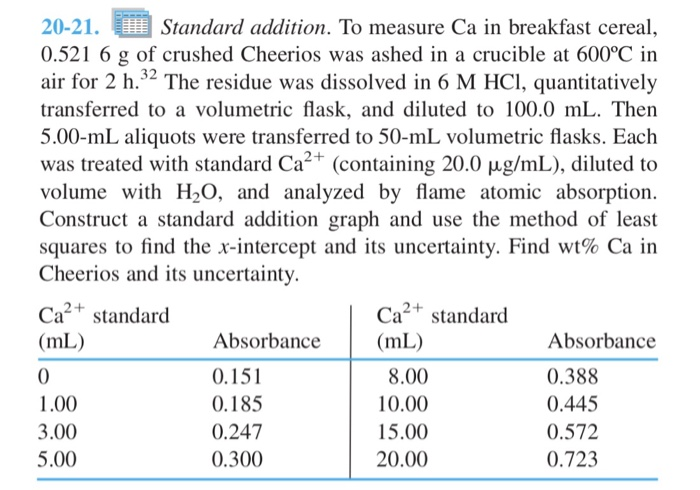
20-21. Standard addition. To measure Ca in breakfast cereal, 0.521 6 g of crushed Cheerios was...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
20-21. Standard addition. To measure Ca in breakfast cereal, 0.521 6 g of crushed Cheerios was ashed in a crucible at 600°C in air for 2 h.32 The residue was dissolved in 6 M HCl, quantitatively transferred to a volumetric flask, and diluted to 100.0 mL. Then 5.00-mL aliquots were transferred to 50-mL volumetric flasks. Each was treated with standard Ca²+ (containing 20.0 µg/mL), diluted to volume with H₂O, and analyzed by flame atomic absorption. Construct a standard addition graph and use the method of least squares to find the x-intercept and its uncertainty. Find wt% Ca in Cheerios and its uncertainty. Ca²+ standard (mL) 0 1.00 3.00 5.00 Absorbance 0.151 0.185 0.247 0.300 Ca²+ standard (mL) 8.00 10.00 15.00 20.00 Absorbance 0.388 0.445 0.572 0.723 20-21. Standard addition. To measure Ca in breakfast cereal, 0.521 6 g of crushed Cheerios was ashed in a crucible at 600°C in air for 2 h.32 The residue was dissolved in 6 M HCl, quantitatively transferred to a volumetric flask, and diluted to 100.0 mL. Then 5.00-mL aliquots were transferred to 50-mL volumetric flasks. Each was treated with standard Ca²+ (containing 20.0 µg/mL), diluted to volume with H₂O, and analyzed by flame atomic absorption. Construct a standard addition graph and use the method of least squares to find the x-intercept and its uncertainty. Find wt% Ca in Cheerios and its uncertainty. Ca²+ standard (mL) 0 1.00 3.00 5.00 Absorbance 0.151 0.185 0.247 0.300 Ca²+ standard (mL) 8.00 10.00 15.00 20.00 Absorbance 0.388 0.445 0.572 0.723
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
To measure Ca in breakfast cereal, 0.521 6 g of crushed Cheerios was ashed in a crucible at 600ºC in air for 2 h.32 The residue was dissolved in 6 M HCl, quantitatively transferred to a...
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
A 50 m3 insulated chamber containing air at 40oC, 100 kPa and R.H. 20% is connected to another 50 m3 insulated chamber containing air at 20oC, 100 kPa and R.H. 100%. The valve is opened and the...
-
Data on the length, in seconds, of a sample of 50 songs by The Beatles are presented in the accompanying data table. Complete parts (a) through (d) below. Click the icon to view the table of song...
-
The three-dimensional (X, Y, Z) coordinates in meters of vertexes A, B, and C in Figure 18.14 are (5412.456, 4480.621, 248.147), (5463.427, 4459.660, 253.121) and (5456.081, 4514.382, 236.193),...
-
Why is there a lovehate relationship between Mexico and the United States?
-
Describe various defenses and remedies available for nonperformance of a contract.
-
Aurora Company spent a total of $102,340 in the current period in one of its production centers. In addition, when the production period began, there was work-in-process inventory in the production...
-
What is a firm's "corporate culture?" List and explain several behaviors that may be attributed to a firm's corporate culture and values system. How can the corporate culture help efforts to promote...
-
Hector Fuego had just been hired by the board of directors to become the new CEO of You Build, Inc. You Build is a 50-year-old building supply company that operates in the southwestern United States....
-
V.S.M. Ltd. is engaged in large scale retail business. From the following informations you are required to forecast their working capital requirements. Projected Annual Sales Rs. 130 lakhs Percentage...
-
Match the terms with their correct definition. Terms 1. Accounts receivable 2. Other receivables 3. Debtor 4. Notes receivable 5. Maturity date 6. Creditor Definitions a. The party to a credit...
-
Cash flow items must be categorized into one of four categories. Identify each item as operating (O), investing (I), financing (F), or non-cash (N). If reported on the statement of cash flows, decide...
-
The following transactions of Plymouth Pharmacies occurred during 2023 and 2024: Journalize the transactions in Plymouths general journal. Explanations are not required. Round to the nearest dollar....
-
Maxwell Furniture Center had accounts receivable of $20,000 at the beginning of the year and $54,000 at year-end. Revenue for the year totaled $116,000. How much cash did the business collect from...
-
How does staff empowerment help in recruitment and staff retention?
-
Katie is working on the income statement for her father's tailor shop. For this month he has revenue of $6,400. His rent on the shop is $600, and he paid $200 in utilities. What is the net income for...
-
(a) What do data breach notification laws require? (b) Why has this caused companies to think more about security?
-
Solutions with a wide range of Hg 2+ concentrations were prepared to calibrate an experimental Hg 2+ ion-selective electrode. For the range 10 -5 < [Hg 2+ ] [ < 10 -1 M, Hg(NO 3 ) 2 was used...
-
The pH of microscopic vesicles (compartments) in living cells can be estimated by infusing an indicator (HIn) into the compartment and measuring the quotient [In-]/[HIn] from the spectrum of the...
-
A 50.00-mL solution containing NaBr was treated with excess AgNO3 to precipitate 0.214 6 g of AgBr (FM 187.772). What was the molarity of NaBr in the solution?
-
The data file \(b r 2\) contains data on 1080 house sales in Baton Rouge, Louisiana, during July and August 2005. The variables are: PRICE (\\($)\), SQFT (total square feet), BEDROOMS (number), BATHS...
-
Explain how and why plots of least squares residuals can reveal heteroskedasticity.
-
How much of an incumbency advantage do winners in U.S. Senate elections enjoy? This issue is examined by Matias D. Cattaneo, Brigham R. Frandsen and Roco Titiunik (2015) "Randomization Inference in...

Study smarter with the SolutionInn App



