Question: Classify the two reactions as ionic or radical. Reaction A: Reaction B: CI: Which statement is true? :FI : CI Reaction A is radical,
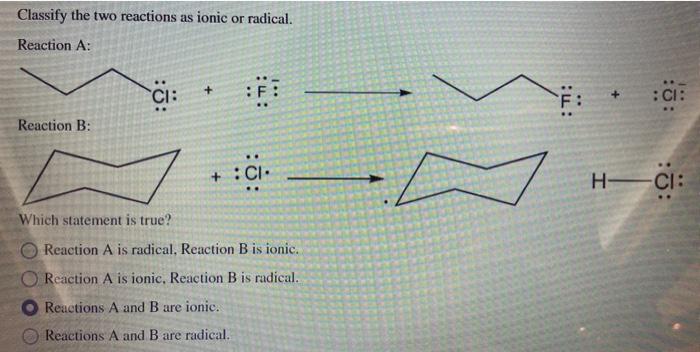
Classify the two reactions as ionic or radical. Reaction A: Reaction B: CI: Which statement is true? :FI : CI Reaction A is radical, Reaction B is ionic.. O Reaction A is ionic, Reaction B is radical. Reactions A and B are ionic. Reactions A and B are radical. T: F : CI: H-CI:
Step by Step Solution
3.45 Rating (148 Votes )
There are 3 Steps involved in it
Reaction A CI F F CI observed was clearly reaction inv... View full answer

Get step-by-step solutions from verified subject matter experts


