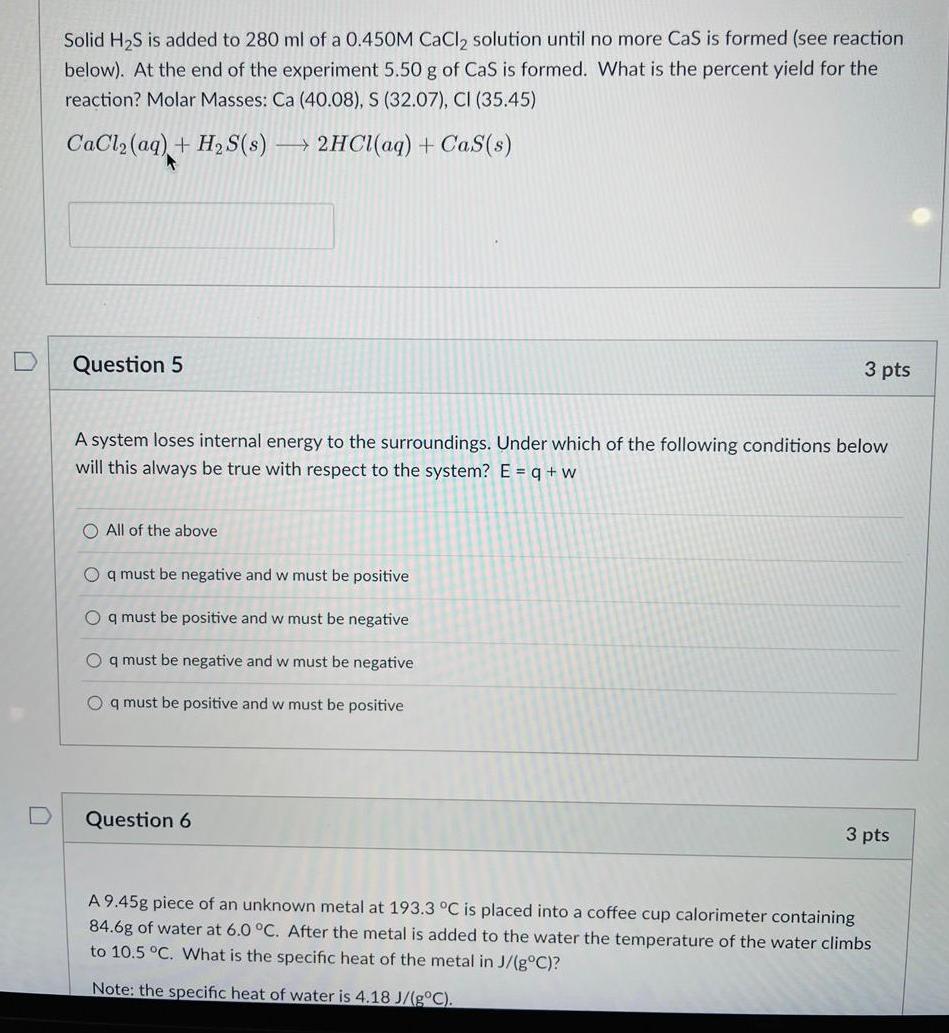
Solid HS is added to 280 ml of a 0.450M CaCl solution until no more CaS...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Solid H₂S is added to 280 ml of a 0.450M CaCl₂ solution until no more CaS is formed (see reaction below). At the end of the experiment 5.50 g of CaS is formed. What is the percent yield for the reaction? Molar Masses: Ca (40.08), S (32.07), CI (35.45) CaCl₂ (aq) + H₂S(s) 2HCl(aq) + Cas(s) A Question 5 A system loses internal energy to the surroundings. Under which of the following conditions below will this always be true with respect to the system? E = q + w O All of the above Oq must be negative and w must be positive Oq must be positive and w must be negative Oq must be negative and w must be negative Oq must be positive and w must be positive 3 pts Question 6 3 pts A 9.45g piece of an unknown metal at 193.3 °C is placed into a coffee cup calorimeter containing 84.6g of water at 6.0 °C. After the metal is added to the water the temperature of the water climbs to 10.5 °C. What is the specific heat of the metal in J/(g°C)? Note: the specific heat of water is 4.18 J/(g°C). Solid H₂S is added to 280 ml of a 0.450M CaCl₂ solution until no more CaS is formed (see reaction below). At the end of the experiment 5.50 g of CaS is formed. What is the percent yield for the reaction? Molar Masses: Ca (40.08), S (32.07), CI (35.45) CaCl₂ (aq) + H₂S(s) 2HCl(aq) + Cas(s) A Question 5 A system loses internal energy to the surroundings. Under which of the following conditions below will this always be true with respect to the system? E = q + w O All of the above Oq must be negative and w must be positive Oq must be positive and w must be negative Oq must be negative and w must be negative Oq must be positive and w must be positive 3 pts Question 6 3 pts A 9.45g piece of an unknown metal at 193.3 °C is placed into a coffee cup calorimeter containing 84.6g of water at 6.0 °C. After the metal is added to the water the temperature of the water climbs to 10.5 °C. What is the specific heat of the metal in J/(g°C)? Note: the specific heat of water is 4.18 J/(g°C).
Expert Answer:
Answer rating: 100% (QA)
4 Percent Yield Calculation The balanced chemical equation for the reaction is CaCl2aqH2Ss2HClaqCaSsCaCl2aqH2Ss2HClaqCaSs To calculate the theoretical ... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
Under which of the following conditions will the first-in, first-out (FIFO) method of process costing produce the same cost of goods manufactured as the weighted-average method? a. When goods...
-
Under which of the following conditions can a disclaimer of opinion never be issued? a. The entitys going- concern problems are highly material and pervasive. b. The entity does not allow the...
-
What is the percent yield for carbon disulfide if the reaction of 40.0 g of carbon produces 36.0g of carbon disulfide?
-
When the government allows private firms to extract minerals offshore or on public lands, two common means of sharing in the profits are bonus bidding and production royalties. The former awards the...
-
What requirements of the Sarbanes-Oxley Act are intended to strengthen the independence of the external auditor?
-
It may be desirable to increase the maximum induced current in the brain tissue. In Fig. P29.73, which time-dependent graph of the magnetic field B in the coil achieves that goal? Assume that...
-
How many chemicals should be reported on in toxic release and national pollutant inventories and polluting emissions registers? What criteria have affected your choice?
-
The adjusted trial balance of Amana Corporation at December 31 shows that sales revenue for the year was $513,000 and other revenue was $37,000. Cost of goods sold for that same period was $256,000,...
-
Cat Bearpaw has just won a sweepstakes and can choose between three prizes. a. $500,000 received 5 years from now. b. $100,000 now, plus $100,000 next year, plus $100,000 in two years. c. $40,000 per...
-
An individuals income varies with his or her age. The following table shows the median income I of males of different age groups within the United States for 2006. For each age group, let the class...
-
8. General Motors has 400,000 shares of common stock outstanding, net income after tax of $1.2 million, retained earnings of $17 million, and total equity of $35 million. What is GM's earnings per...
-
1) Compare the variables in the Binomial model with those in the black-schooled model. Discuss which ones differ between the two models. Explain how this variable is presented in the each of these...
-
what is the most efficient way to see the sources and targets related to a transaction?
-
What is the prediction of the CAPM with respect to the expected return on any security? Why is the variance of a portfolio of internationally diversified stocks likely to be lower than the variance...
-
Lagoon Plc's Directors are planning to instigate the removal of its auditors, KPNG due to their inability to deliver "quality audit works". However, the Audit Manager has insisted that this move by...
-
Firms A and B are identical in their business activity. Both have following annual data: EBITDA 5,000.00; Depreciation=1,000.00 ; TAX=20% ; OWC=0 and CAPEX=1,000.00 Firm A has no debt; Firm B...
-
a new two contract for the building was signed with a 10.00% increase from the prior year insurance expenses will hold steady due to contract agreement travel/entertainment will be reduced by 7.00%...
-
One Way Cellular accountants have assembled the following data for the year ended September 30, 2014: Prepare the operating activities section using the indirect method for One Way Cellulars...
-
Abigail (Abby) Boxer is a single mother working as a civilian accountant for the U.S. Army. Her Social Security number is 676-73-3311 and she lives at 3456 Alamo Way, San Antonio, TX 78249. Helen,...
-
In the 2012 tax year, Michelle paid the following amounts relating to her 2010 tax return: Tax deficiency..........................................$5,000 Negligence...
-
Calculate the amount of the child and dependent care credit allowed for 2012 in each of the following cases, assuming the taxpayers had no income other than the stated amounts. a. William and Carla...
-
Valley Bleu Lte reported the following current accounts at December 31, 2019 (amounts in thousands): During 2020, Valley Bleu completes these transactions: - Used inventory of \(\$ 3,800\) - Sold...
-
Tidy Car, Inc.. provies mohile detailing to its customers. The Income Statement for the month ended January 31, 2020, the Balance Sheet for December 31, 2019, and details of postings to the Cash...
-
Journalize the adjusting entry needed on December 31, 2020, the end of the current accounting period, for each of the following independent cases affecting Callaway Corp. Include an explanation for...

Study smarter with the SolutionInn App


