Complete each of the following nuclear decay reactions. Mg Al + 134 210 Rn ...
Fantastic news! We've Found the answer you've been seeking!
Question:
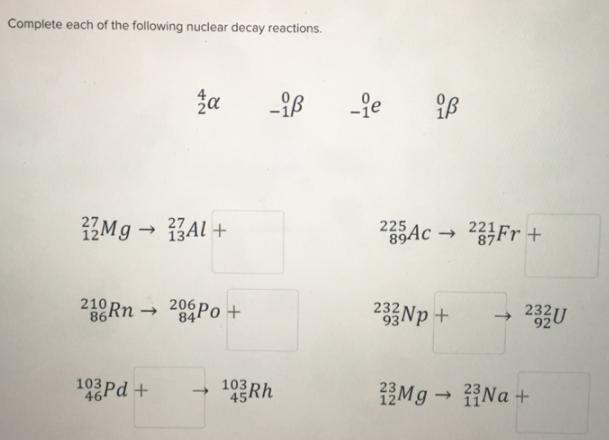
Transcribed Image Text:
Complete each of the following nuclear decay reactions. Mg → Al + 134 210 Rn → 86 za 103 Pd+ 464 206 Po+ 84 103 -B -ie 225 89Ac iß 233Np + 223Fr + ->> 232 U Mg → Na + Complete each of the following nuclear decay reactions. Mg → Al + 134 210 Rn → 86 za 103 Pd+ 464 206 Po+ 84 103 -B -ie 225 89Ac iß 233Np + 223Fr + ->> 232 U Mg → Na +
Expert Answer:
Answer rating: 100% (QA)
Answer is given in the below image 1 1 27 mg 27 Al ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
Each of the following nuclei undergoes either beta decay or positron emission. Predict the type of emission for each: (c) Iodine-120 (d) silver-102. }H,
-
Complete each of the reactions given in Fig. P20.49 by giving the principal organic product(s). Give the reasons for your answers. H,C KOH PhCH Cl acid HO,C CO,H+ethylene glycolheolym) CH + Hg(OAch...
-
The enthalpy change for each of the following reactions was calculated using bond energies. The bond energies of XO, YO, and ZO are all equal. XX + O=O XOOX; H = 275 kJ YY + O=O YOOY; H = +275 kJ...
-
Problem A (20 points): Loco Farms Company sold 36,000 units of its only product and incurred a $18,672 loss (ignoring taxes) for the current year as shown here. During a planning session for year...
-
A company operates two large plants. In one plant, a random sample of 150 employees revealed that 23 had had workplace accidents during the past year. In the other plant, a random sample of 125...
-
Create a star schema for this case study. How did you handle the time dimension? Fitchwood Insurance Company, which is primarily involved in the sale of annuity products, would like to design a data...
-
Integral balances can also be used for heat transfer in a turbulent-flow boundary layer if a form for the velocity profile is assumed. A common form is the 1/7th-power law:...
-
Jane James owns an appliance store. She normally receives $50,000 worth of appliances per month. She does not like to owe people money and always pays her bills on the day she receives the invoice....
-
By increasing the areas of the zinc and copper plates in the Daniell cell, we can increase the amperage of the current flowing through the circuit without affecting the cell's voltage. (a) Why does...
-
Lee, Danny and Sue were childhood friends. They loved horses and dreamed of one day owning a horse who won the huge prize money in the famous annual Melbourne Cup horse race. To fulfill this dream,...
-
Write a program for the following problems. It is recommended that the programs should be written and run on Ubuntu environment using C/C++. However, students can also use other tools to accomplish...
-
Musashi Ltd. commenced operations on January 1, 2016. on that day, it bought: Equipment costing 3.6 million, with a useful life of 3 years. Furniture and fittings costing 2.4 million, with a useful...
-
An acre (43,560 SF) of land in the Central Business District (CBD) is being used as an open parking lot. The land currently brings an operating cashflow of $28/SF/year, paid in arrears, which is...
-
Some companies like Facebook Inc. have two stages of growth, a fast rate for the next few years and then a slower rate for years after this. The valuation of a company like this will have two parts;...
-
Consider a one-period economy with two times, 0 and T. There are S = 3 states and N = 2 securities with payoffs and prices p = [10, 10]'. D = ( 10 9 10 10 10 11 (a) Does this market satisfy the Law...
-
Fox Manufacturing Company Income Statement for the Year Ended December 31, 2021 Sales revenue Less: Cost of goods sold Gross profits Less: Operating expenses General and administrative expenses...
-
Ship taking on water: A ship collides with an iceberg at t = 0. One of the compartments of the ship, which is initially dry, starts to fill with water at a rate at, measured in liters per second,...
-
a) Calculate the goodwill that was paid by Major Ltd on the acquisition of Minor Ltd. [10 marks] b) Prepare the consolidated statement of financial position for Major Ltd at 31 July 20X8. [30 marks]...
-
Three cations, Ni2+,Cu2+, and Ag +, are separated using two different precipitating agents. Based on Figure 17.23, what two precipitating agents could be used? Using these agents, indicate which of...
-
Calculate the minimum pH needed to precipitate Mn(OH)2 so completely that the concentration of is less than 1 per liter [1 part per billion (ppb)].
-
Consider the reaction 3 CH4 (g) C3H8(g) + 2 H2(g). (a) Using data from Appendix C, calculate G at 298 K. (b) Calculate G at 298 K if the reaction mixture consists of 40.0 atm of CH4, 0.0100 atm of...
-
Factor (smart beta) strategy ETFs are least likely to be used by investors: A. to modify portfolio risk. B. for tactical trading purposes. C. to seek outperformance versus a benchmark.
-
Which of the following statements regarding applications of ETFs in portfolio management is correct? A. Equity ETFs tend to be more active than fixed-income ETFs. B. The range of risk exposures...
-
Which of Stosurs statements regarding ETF mechanics is correct? A. Statement 1 B. Statement 2 C. Statement 3 Howie Rutledge is a senior portfolio strategist for an endowment fund. Rutledge meets with...

Study smarter with the SolutionInn App


