Complete the Lewis structure of the compound shown below and indicate which of the following statements are
Question:
Complete the Lewis structure of the compound shown below and indicate which of the following statements are true.
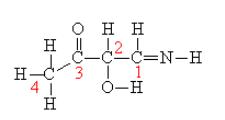
The C-N bond is formed from overlap of an sp3 hybrid orbital from the carbon atom with an sp2 hybrid orbital from the nitrogen atom.
true false The C-2 and C-3 atoms cannot rotate about the internuclear axis between the two atoms since the σ bond would break.
true false There are four π bonds in this molecule.
true false There are three lone pairs of electrons in the complete Lewis structure.
true false An sp2 hybrid orbital on C-1 overlaps with an sp3 hybrid orbital on C-2 to form the sigma bond between C-1 and C-2.
true false The π bond between C-3 and O is formed by overlap of unhybridized p atomic orbitals.
true false There are eleven σ bonds in this molecule.

Income Tax Fundamentals 2015
ISBN: 9781305177772
33rd edition
Authors: Gerald E. Whittenburg, Martha Altus-Buller, Steven Gill




