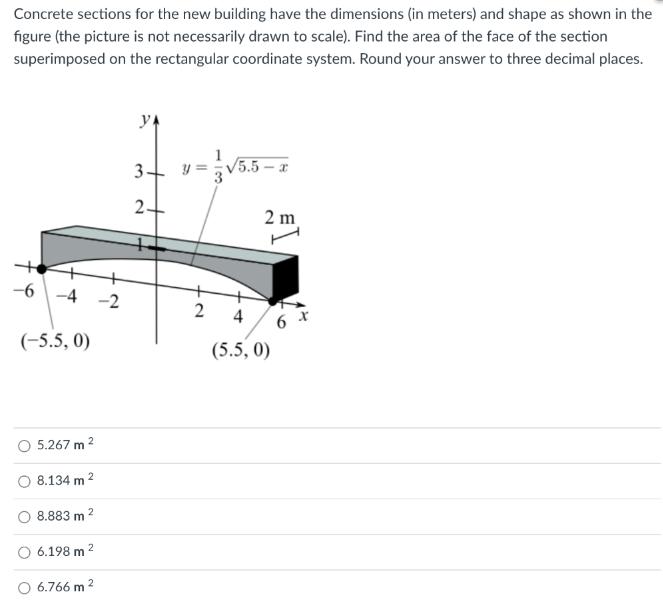
Concrete sections for the new building have the dimensions (in meters) and shape as shown in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Concrete sections for the new building have the dimensions (in meters) and shape as shown in the figure (the picture is not necessarily drawn to scale). Find the area of the face of the section superimposed on the rectangular coordinate system. Round your answer to three decimal places. -4-2 (-5.5, 0) 2 5.267 m 8.134 m 2 8.883 m ² 6.198 m 2 6.766 m ² ya 3- 2+ y = √5.5-x 2 2 m 4 (5.5, 0) 6 x Concrete sections for the new building have the dimensions (in meters) and shape as shown in the figure (the picture is not necessarily drawn to scale). Find the area of the face of the section superimposed on the rectangular coordinate system. Round your answer to three decimal places. -4-2 (-5.5, 0) 2 5.267 m 8.134 m 2 8.883 m ² 6.198 m 2 6.766 m ² ya 3- 2+ y = √5.5-x 2 2 m 4 (5.5, 0) 6 x
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
Overview: In the first milestone of your final project, you will determine and classify the costs necessary for opening your business. This is a critical step that must be done correctly for you to...
-
Suppose x has a distribution with = 69 and = 17. (a) If random samples of size n = 16 are selected, can we say anything about the x distribution of sample means? Yes, the x distribution is normal...
-
Find the area between the following curves. Truncate your answer to three decimal places (2.5-2.500, 2.7856-2.785) y 4 and y 2x
-
1. Suppose that a stoichiometric mixture of isooctane (C8H18) and air is burned in an engine and then the fuel is changed to 10% (by liquid volume) ethanol and 90% by liquid volume isooctane. If the...
-
The following data pertain to Aurora Electronics for the month of February. Required: Compute the sales-price and sales-volume variances forFebruary. Static Budget Actual 9,000 $120,000 $103,500...
-
A cylindrical specimen of a brass alloy having a length of 100 mm (4 in.) must elongate only 5 mm (0.2 in.) when a tensile load of 100,000 N (22,500 lbf) is applied. Under these circumstances, what...
-
On January 1,2010, you borrowed $10,000 on a five-year, 8% note payable. At December 31,2010, you should record a journal entry that includes which of the following? a. Note Payable of $10,000 b....
-
Discuss the risks depicted by the fixed asset system flowchart for Problem 10. Describe the internal control improvements to the system that are needed to reduce theserisks. Vendor User Department...
-
How Do Intimate Apparel makers pick their fabric and what fabric properties would be needed to satisfy your customers and their specific needs?
-
The following transactions of Jacks Auto Supply occurred in November (Balances as of November 1 are given for general ledger and accounts receivable ledger accounts: Danielson $1,100 Dr.; Wallace...
-
Moving Up Chemical Productions Pty Ltd ('the company') holds an environment protection licence ('EPL') under the Protection of the Environment Operations Act 1997 (NSW) ('the POEO Act') in relation a...
-
Youve no doubt heard of 5-Hour Energythe small red bottles filled with an energy-infused elixir, located alongside the candy and gum near the checkout at most grocery and convenience stores. Chances...
-
Review the strategies available to companies when determining how to best distribute their products: intensive, exclusive, and selective distribution. Select five products (in more than one category)...
-
Evaluate the sales promotion activities of a grocery store in your area. What consumer sales promotions is it using? Is it successful at reaching its target market with effective promotions?
-
Many companies today are looking to do more than just good deeds. More and more, companies are making environmental sustainability a part of their core missions. Research companies that make...
-
Market research is critical for companies that want to grow their markets. Companies can hire outside market research firms or create their own internal market research positions or teams. There are...
-
What is the result of each of the following statements? [In the event a list is an answer, just write the list not the annotated version - Example: (a b c) NOT (list a b c) a. (car' (a (b c d))) b....
-
In Exercises 1-2, rewrite each verbal statement as an equation. Then decide whether the statement is true or false. Justify your answer. 1. The logarithm of the difference of two numbers is equal to...
-
Tartaric acid, H2C4H4O6, has two acidic hydrogens. The acid is often present in wines and precipitates from solution as the wine ages. A solution containing an unknown concentration of the acid is...
-
(a) What are the CCC bond angles in diamond? (b) What are they in graphite?
-
In the year 2007, an estimated amount of 31 billion tons of carbon dioxide (CO2) was emitted worldwide due to fossil fuel combustion and cement production. Express this mass of CO2 in grams without...
-
For an ideal solution, isotherm on an enthalpy-concentration diagram will be (a) Parabola (b) Hyperbola (c) Sine curve (d) Straight line
-
For a reversible process, change in entropy of the system (a) Approaches to zero (b) Increases (c) Decreases (d) Remains constant
-
For a multi-component system, the chemical potential is equivalent to (a) Molar free energy (b) Molar concentration difference (c) Molar free energy change (d) Partial molar free energy

Study smarter with the SolutionInn App



