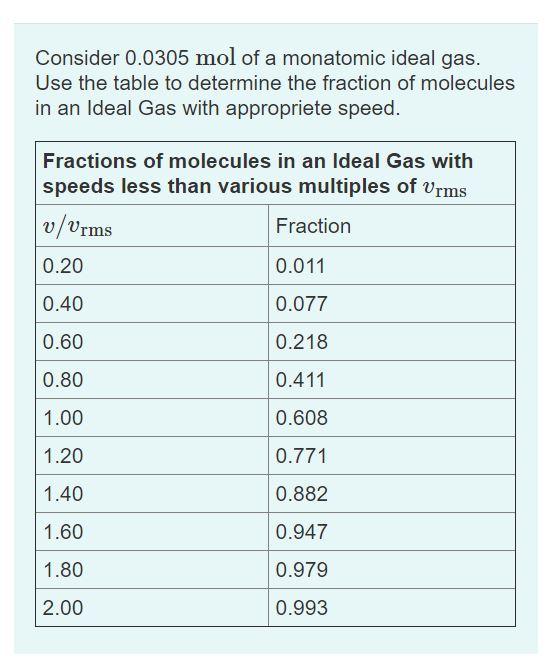
Consider 0.0305 mol of a monatomic ideal gas. Use the table to determine the fraction of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider 0.0305 mol of a monatomic ideal gas. Use the table to determine the fraction of molecules in an Ideal Gas with appropriete speed. Fractions of molecules in an Ideal Gas with speeds less than various multiples of vrms v/Vrms Fraction 0.20 0.011 0.40 0.077 0.60 0.218 0.80 0.411 1.00 0.608 1.20 0.771 1.40 0.882 1.60 0.947 1.80 0.979 2.00 0.993 Part A How many of the atoms have speeds that are within 20% of the rms speed? Express your answer in amount of atoms. ΑΣφ atoms %3D Consider 0.0305 mol of a monatomic ideal gas. Use the table to determine the fraction of molecules in an Ideal Gas with appropriete speed. Fractions of molecules in an Ideal Gas with speeds less than various multiples of vrms v/Vrms Fraction 0.20 0.011 0.40 0.077 0.60 0.218 0.80 0.411 1.00 0.608 1.20 0.771 1.40 0.882 1.60 0.947 1.80 0.979 2.00 0.993 Part A How many of the atoms have speeds that are within 20% of the rms speed? Express your answer in amount of atoms. ΑΣφ atoms %3D
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
The temperature of 2.5 mol of a monatomic ideal gas is 350 K. The internal energy of this gas is doubled by the addition of heat. How much heat is needed when it is added at (a) Constant volume and...
-
A 1.00-mol sample of a monatomic ideal gas is taken through the cycle shown in Figure P22.65. At point A, the pressure, volume, and temperature are Pi , Vi , and Ti , respectively. In terms of R and...
-
The temperature of 1.00 mol of a monatomic ideal gas is raised reversibly from 300 K to 400 K, with its volume kept constant. What is the entropy change of the gas?
-
Lets assume the client in the fact pattern in this weeks discussion board did not want to purchase the mothers house (in other words, forget about only those facts) and now has been appointed as the...
-
In 1908, W. S. Gosset published the article The Probable Error of a Mean (Biometrika, Vol. 6, pp. 125). In this pioneering paper, written under the pseudonym Student, Gosset introduced what later...
-
Using the data in Table 12.5, do the following: (a) Determine the flexural strength for nonporous MgO assuming a value of 3.75 for n in Equation 12.10. (b) Compute the volume fraction porosity at...
-
Explain each of the relationships outlined in the quadrants in Figure 10.1 (page 239). Detail the actions that would lead to a favourable change in direction in each of the four relationships...
-
Model 99 Hotels is considering the construction of a new hotel for $80 million. The expected life of the hotel is 20 years with no residual value. The hotel is expected to earn revenues of $15...
-
Training is an expensive undertaking for any organisation, yet its benefits may not be immediate and obvious. Explain few reasons why organizations undertake employee training despite its limitations?
-
An infinite slab of dielectric material dielectric constant x is placed in a unif electric field of magnitude E. The fiel perpendicular to the surface of the slab. field inside the slab is (A) E E%E0...
-
Compute and report the Macaulay duration for each of the 12 US bonds. Report this in three 24 tables, where the first row corresponds to the bond number and the second to the Macaulay duration. The...
-
Prepare the following a. The Statement of Profit and Loss for the year ending December 31, 2020 ( 27 marks ) b. The Statement of Financial Position at at December 31, 2020 The following trial balance...
-
Discuss how you will use what we learned in the course in a professional setting. What did you learn that you will find particularly helpful in your future? What websites or sources did you find...
-
The following events took place for Rushmore Biking Inc. during February, the first month of operations as a producer of road bikes: Purchased $600,200 of materials. Used $516,200 of direct...
-
20 You purchased a share of stock for $20. One year later you received $1 as a dividend and sold the share for $24. What was your holding-period return? A 20% B 50% C 25% D 40% E None of the options...
-
How do societies define and regulate deviance, and what role do formal and informal mechanisms of social control play in maintaining societal cohesion ?
-
Following are the current asset and current liability sections of the balance sheets for Freedom Incorporated at January 31, 2023 and 2022 (in millions): January 31, January 31, 2023 2022 Current...
-
Anna, a high school counselor, devised a program that integrates classroom learning with vocational training to help adolescents at risk for school dropouts stay in school and transition to work...
-
Multiple-Concept Example offers useful background for problems like this. A cylinder is rotating about an axis that passes through the center of each circular end piece. The cylinder has a radius of...
-
A converging lens (f = 12.0 cm) is located 30.0cm to the left of a diverging lens (f = -6.00 cm). A postage stamp is placed 36.0 cm to the left of the converging lens. (a) Locate the final image of...
-
A microscope for viewing blood cells has an objective with a focal length of 0.50 cm and an eyepiece with a focal length of 2.5cm. The distance between the objective and eyepiece is 14.0cm. If a...
-
Net sales, net income, and total assets for Azbell Shipping, Inc., for a five-year period follow: {Requirements} 1. Compute trend percentages for each item for 2017 through 2020. Use 2016 as the base...
-
The financial statements of Adventure News, Inc., include the following items: {Requirements} 1. Compute the following ratios for 2020 and 2019 a. Current ratio b. Quick (acid-test) ratio c....
-
You have been asked to evaluate two companies as possible investments. The two companies, Norfolk Industries Inc. and Strafford Crystal Limited, are similar in size. Assume that all other available...

Study smarter with the SolutionInn App