Consider a buffer composed of aqueous solutions of HCN and KCN. What is the net ionic...
Fantastic news! We've Found the answer you've been seeking!
Question:
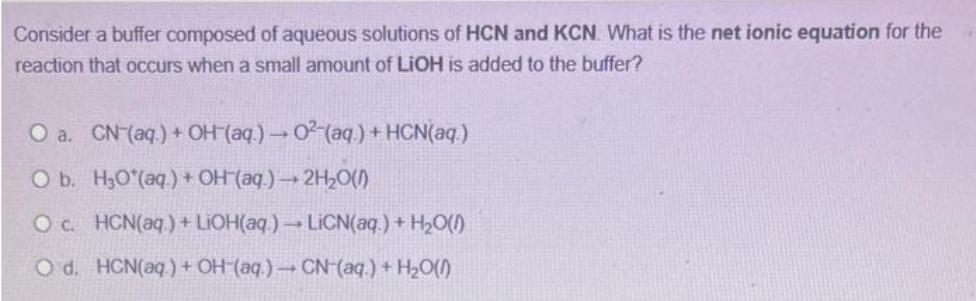
Transcribed Image Text:
Consider a buffer composed of aqueous solutions of HCN and KCN. What is the net ionic equation for the reaction that occurs when a small amount of LiOH is added to the buffer? O a. CN- (aq) + OH(aq) → 02(aq) + HCN(aq) O b. H₂O*(aq) + OH (aq) → 2H₂O(1) O c. HCN(aq) + LiOH(aq.) → LiCN(aq) + H₂O(l) O d. HCN(aq) + OH-(aq.) → CN-(aq) + H₂O(1) Consider a buffer composed of aqueous solutions of HCN and KCN. What is the net ionic equation for the reaction that occurs when a small amount of LiOH is added to the buffer? O a. CN- (aq) + OH(aq) → 02(aq) + HCN(aq) O b. H₂O*(aq) + OH (aq) → 2H₂O(1) O c. HCN(aq) + LiOH(aq.) → LiCN(aq) + H₂O(l) O d. HCN(aq) + OH-(aq.) → CN-(aq) + H₂O(1)
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provide... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
What is the net ionic equation for the following molecular equation? Hydrofluoric acid, HF, is a molecular substance and a weak electrolyte. HF(aq) + KH() KFaq) + .()
-
(a) Write the net ionic equation for the reaction that occurs when a solution of hydrochloric acid (HCl) is mixed with a solution of sodium formate (NaCHO 2 ). (b) Calculate the equilibrium constant...
-
Select the net ionic equation for the reaction between sodium chloride and mercury(I) nitrate. 2NaCl( aq ) + Hg 2 (NO 3 ) 2 ( aq ) 2NaNO 3 ( aq ) + Hg 2 Cl 2 ( s ) A. Na + ( aq ) + NO 3 ( aq ) ...
-
Campione Manufacturing acquired an 80% interest in DaLuca Distributors, a foreign corporation established on November 1, 2006, for 650,000 foreign currency units (FC). Campione acquired its 80%...
-
Justin owns a small factory that produces buttons for garment industry. He wants to buy a new machine and has applied to a bank for financing. The loan officer has asked for operating statements for...
-
A local news survey asked 500 randomly sampled Los Angeles residents which shipping carrier they prefer to use for shipping holiday gifts. The table below shows the distribution of responses by age...
-
A uniform magnetic field exists in a circular area. A particle carrying charge \(q=5.0 \mathrm{mC}\) is placed in the field a distance \(r_{\mathrm{p}}=20 \mathrm{~mm}\) from the center of the...
-
The Giant Farmer Company processes food for sale in discount food stores. ft has two plants: one in Chicago and one in Houston. The company also operates warehouses in Miami. Florida: Denver,...
-
11 12 13 14 Height of a plant is regulated by: a) DNA which is directly influenced by growth hormone. b) Genes which regulate the proteins directly. Growth hormones under the influence of the enzymes...
-
1. Name at least three ways that Shu could automate her asset management. Suggest at least one option for retirement savings, general savings, and general convenience. 2. What major factors should...
-
QUESTION 14 1. AC Electric just paid a $2.10 per share annual dividend. The firm pledges to increase its dividend by 2.4 percent for the next 5 years and then maintain a constant 2 percent rate of...
-
Bridge Builders Ltd undertook a contract to build a pedestrian footbridge for a fixed price of 400,000 during the period from May Year 1 to July Year 2. This table sets out transactions up to the...
-
A product is manufactured in a continuous process carried on successively in two departments, Assembly and Finishing. In the production process, materials are added to the product in each department...
-
Set out below are three job cost records. Prepare the work-in-progress control account in the general ledger which represents the total of these three separate records. Month of April Direct...
-
How is the profit on an incomplete contract calculated.
-
Refinery Ltd buys crude oil which is refined, producing liquefied gas, oil and grease. The cost of crude oil refined in the past year was 105,000 and the refining department incurred processing costs...
-
The following data are available for the current year for C Company. C Company uses a 365-day year when computing ratios. Annual credit sales = $18 million Cost of goods sold = $12 million Inventory...
-
d. The characteristic equation of a control system is given by s+2s+8s+12s+20s+16+16=0. Determine the number of the roots of the equation which lie on the imaginary axis of s-plane
-
(a) Explain why BrF4- is square planar, whereas BF4- is tetrahedral. (b) How would you expect the H-X-H bond angle to vary in the series H2O, H2S, H2Se? Explain.
-
(a) What do you expect for the sign of S in a chemical reaction in which two moles of gaseous reactants are converted to three moles of gaseous products? (b) For which of the processes in Exercise...
-
Explain the structure of the periodic table-two columns on the left, a block of ten for the transition metals, a block of six on the right, and a pair of 14-member rows below, with reference to the...
-
Problem Decide whether the sampling method is independent or dependent. Then determine whether the response variable is qualitative or quantitative. (a) Joliet Junior College decided to implement a...
-
Using the data from Table 2, construct a \(95 \%\) confidence interval estimate of the mean difference, \(\mu_{d}\). By Hand Approach Step 1 Compute the differenced data. Because the sample size is...
-
In the Spacelab Life Sciences 2 payload, 14 male rats were sent to space. Upon their return, the red blood cell mass (in milliliters) of the rats was determined. A control group of 14 male rats was...

Study smarter with the SolutionInn App


