Consider a first-order irreversible reaction that occurs in a packed bed reactor where CA has units...
Fantastic news! We've Found the answer you've been seeking!
Question:
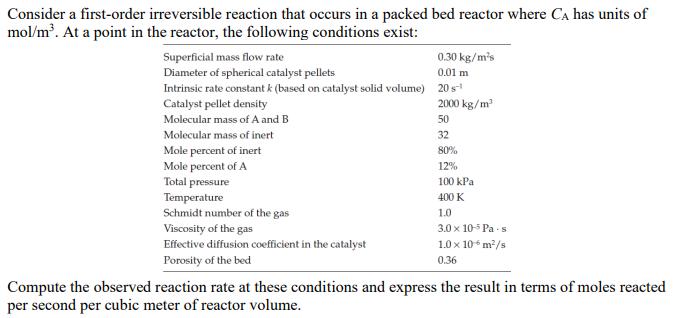
Transcribed Image Text:
Consider a first-order irreversible reaction that occurs in a packed bed reactor where CA has units of mol/m. At a point in the reactor, the following conditions exist: Superficial mass flow rate Diameter of spherical catalyst pellets 0.30 kg/ms Intrinsic rate constant k (based on catalyst solid volume) 20s- Catalyst pellet density Molecular mass of A and B Molecular mass of inert 0.01 m 2000 kg/m 50 32 80% 12% Mole percent of inert Mole percent of A Total pressure Temperature Schmidt number of the gas Viscosity of the gas Effective diffusion coefficient in the catalyst Porosity of the bed 100 kPa 400 K 1.0 3.0 x 10-5 Pas 1.0 10 6 m/s 0.36 Compute the observed reaction rate at these conditions and express the result in terms of moles reacted per second per cubic meter of reactor volume. Consider a first-order irreversible reaction that occurs in a packed bed reactor where CA has units of mol/m. At a point in the reactor, the following conditions exist: Superficial mass flow rate Diameter of spherical catalyst pellets 0.30 kg/ms Intrinsic rate constant k (based on catalyst solid volume) 20s- Catalyst pellet density Molecular mass of A and B Molecular mass of inert 0.01 m 2000 kg/m 50 32 80% 12% Mole percent of inert Mole percent of A Total pressure Temperature Schmidt number of the gas Viscosity of the gas Effective diffusion coefficient in the catalyst Porosity of the bed 100 kPa 400 K 1.0 3.0 x 10-5 Pas 1.0 10 6 m/s 0.36 Compute the observed reaction rate at these conditions and express the result in terms of moles reacted per second per cubic meter of reactor volume.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
After deducting capital allowances for the year to 31 December 2019, the tax written down values of a company's plant and machinery were as follows: Calculate the capital allowances available for the...
-
TRM Consulting Services currently has the following capital structure: Source Book Value Quantity Common Stock........................ $ 8,500,000..................... 350,000 Preferred...
-
1. Why do you think InnovationCo used the specific standardization and knowledge networking approach in IHRM? What other options did it have? 2. What are the advantages and disadvantages associated...
-
This case stems from a rocky relationship between two Texans. Lawrence Shipley was the president of Shipley Do-Nut Flour & Supply Co., and Andrea Vasquez was a substitute school teacher in the...
-
The completed financial statement columns of the worksheet for Fleming Company are shown below. Instructions (a) Prepare an income statement, an owners equity statement, and a classified balance...
-
What is the central concept in urban economics that says people/businesses being physically next together increases overall productivity? Why does it increase productivity?
-
Lee, Danny and Sue were childhood friends. They loved horses and dreamed of one day owning a horse who won the huge prize money in the famous annual Melbourne Cup horse race. To fulfill this dream,...
-
Let f(x, y) = 5 (x - x2y) if 0< x < 1 and 0 < y < 1 and zero otherwise. Find E(XY).
-
The purpose of inoculation is A . . To clean the casting B . . To decrease the melting temperature of a cast metal C . . To alter the chemical composition of a cast metal D . . To modify the...
-
Modern gear both profile is given involute shape because (A) this is a very easy curve for manufacturing (B) sliding does not takes place anywhere on meshing teeth (C) involute is the only profile...
-
Given the following cross-sections (with units in mm): a) b) t = 2 X b = 20 d) h= 20 b = 30 T Y t = 2 a 10 c = 30 t = 1.5 b=15 X h=20 t = 1.5 b=15 X Figure 3: Cross-sections for problem 2. 2 0=6 a =...
-
The imagelhlkur shows a cylindrical rod that is composed of two metals, aluminum and brass. A rigid plate is placed on the end and pushed down by a force P. The spring coefficients of the materials...
-
A green fifth-band on a precision resistor indicates a tolerance value of : A 2% B 0.25% 1% 0.5%
-
1.Describe in a general way equilibrium equations to find reactions in supports. Denote neccessary intervals by corresponding characters. M 2M
-
On January 1, 2018, Khalid Ltd., which follows IAS 17, entered into an eight-year lease agreement for three dryers. Annual lease payments for the equipment are $28,500 at the beginning of each lease...
-
Suggest combinations of ideal reactors to model the real reactors given in Problem P16-2b(b) for either E(), E(t), F(), F(t), or (1 F()).
-
AWFOSS5 View the CSB video (https://www.youtube.com/watch?v=-_ZLQkn7X-k) and then research the conclusion as to whether or not the safety analysis of the incident is complete.
-
Prepare a list of safety considerations for designing and operating chemical reactors. What would be the first four items on your list? For example, what safety concerns would you have for operating...
-
Around $500 billion in U.S. Treasuries are traded each day and many countries hold U.S. Treasuries, as they are considered a very safe investment. For example, as of October 2018, Japan held $1,018.5...
-
You are working at the campus bookstore earning $9.00 per hour. Your manager tells you that in the upcoming year you will get a 2% raise. How does your real wage change if inflation next year is 1%,...
-
In 2018, the federal government spent $4.1 trillion and brought in revenue of $3.3 trillion. a. Is this a budget deficit or surplus, and what is its size? b. If net government debt in 2018 was $15.8...

Study smarter with the SolutionInn App


