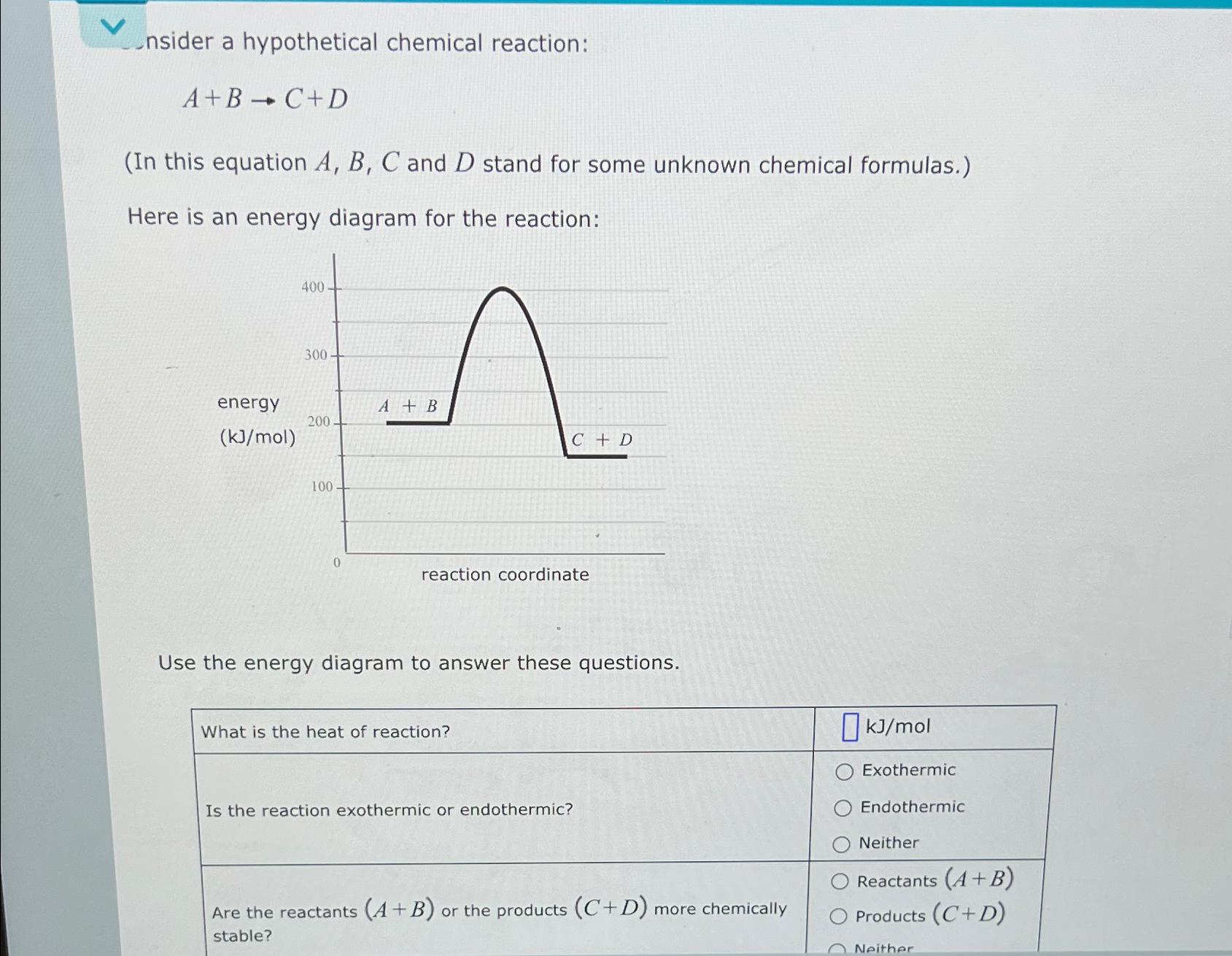
Consider a hypothetical chemical reaction: A+B C+D - (In this equation A, B, C and D...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider a hypothetical chemical reaction: A+B C+D - (In this equation A, B, C and D stand for some unknown chemical formulas.) Here is an energy diagram for the reaction: energy (kJ/mol) 400- 300 200- 100 0 A + B C + D reaction coordinate Use the energy diagram to answer these questions. What is the heat of reaction? Is the reaction exothermic or endothermic? Are the reactants (A + B) or the products (C+D) more chemically stable? kJ/mol Exothermic Endothermic Neither Reactants (A+B) Products (C+D) Neither Consider a hypothetical chemical reaction: A+B C+D - (In this equation A, B, C and D stand for some unknown chemical formulas.) Here is an energy diagram for the reaction: energy (kJ/mol) 400- 300 200- 100 0 A + B C + D reaction coordinate Use the energy diagram to answer these questions. What is the heat of reaction? Is the reaction exothermic or endothermic? Are the reactants (A + B) or the products (C+D) more chemically stable? kJ/mol Exothermic Endothermic Neither Reactants (A+B) Products (C+D) Neither
Expert Answer:
Answer rating: 100% (QA)
The energy differential between the reactants and products can be u... View the full answer

Related Book For 

Microeconomics An Intuitive Approach with Calculus
ISBN: 978-0538453257
1st edition
Authors: Thomas Nechyba
Posted Date:
Students also viewed these chemical engineering questions
-
Consider the QHIC data in Figure 13.21 (page 556). When we performed a regression analysis of these data by using the simple linear regression model, plots of the model's residuals versus x (home...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Carrie Carson: Carrie is a 60-year-old tai chi instructor living in Santa Fe, New Mexico. For many years, she practiced in Hollywood, and because of an acting background, she developed a clientele...
-
Portable Apps for Forensics Investigations Portable apps and bootable Linux distributions are growing in popularity. Not only are they highly transportable via thumb drive, but they also run faster...
-
The Chelsea location of Family Supermarket (FS), a chain of small neighbourhood grocery stores, is preparing its activity-based budget for January 2013. FS has three product categories: soft drinks,...
-
A 9.0-nm-thick cell membrane undergoes an action potential that follows the curve in the table on page 748 . What is the strength of the electric field inside the membrane just before the action...
-
How do accounting principles affect financial statement analysis?
-
1. The manufacturer-recommended pressure of air in the tires of US passenger cars is typically 30 psi (gauge). (a) Find the absolute pressure of the air inside tires and express it in mmHg, kPa, and...
-
Acrylic bone cement is commonly used in total joint replacement to secure the artificial joint. Data on the force (measured in Newtons, N) required to break a cement bond under two different...
-
has the domestic domestic supply and for a good. According to the table, the domestic price of the good is The table demand schedules
-
Ingram Office Supplies, Inc., buys on terms of 2/15, net 50 days. It does not take discounts, and it typically pays on time, 50 days after the invoice date. Net purchases amount to $300,000 per year....
-
Kwashiorkor is a severe malnutrition common in children who do not get enough protein in their diet. It can lead to swelling in the abdomen. What is this swelling known as and why would it occur?
-
For what minimum period must a self-employed taxpayer retain business documents supporting her federal tax return?
-
Malcolm McBeth is remodeling his kitchen and the proposal from Ace Corp is at a cost of $100,000. The salesperson promises annual savings as follow and Malcolm has a cost of capital of 10 percent and...
-
would an S-Corp owner/employee rather take money out of his/her S-Corp as salary or as distributions? Be specific in your answer and provide an explanation. What is the tax accountant's role in...
-
Two word-wide unsigned integers are stored at the physical memory address 00A0016 and 00A0216, respectively. Write an instruction sequence that computes and stores their sum, difference, product and...
-
5. How much would you need to deposit in an account now in order to have $5,000 in the account in 5 years? Assume the account earns 2% interest compounded monthly. 10. You deposit $300 each month...
-
Suppose you and I have the same homothetic tastes over x1 and x2, and our endowments of the two goods are EM = (eM1, eM2) for me and EY = (eY1, eY2) for you. A: Suppose also, again as in exercise...
-
Demand for Charities and Tax Deductibility: One of the ways in which government policy supports a variety of activities in the economy is to make contributions to those activities tax deductible. For...
-
After you graduate, you get a job in a small city where you have taken your sisters offer of living in her apartment. Your job pays you $20 per hour and you have up to 60 hours per week available....
-
A surface with \(N_{0}\) adsorption centers has \(N\left(\leq N_{0}ight)\) gas molecules adsorbed on it. Show that the chemical potential of the adsorbed molecules is given by \[ \mu=k T \ln...
-
Show that for a system in the grand canonical ensemble \[ \{\overline{(N E)}-\bar{N} \bar{E}\}=\left(\frac{\partial U}{\partial N}ight)_{T, V} \overline{(\Delta N)^{2}} \]
-
Study the state of equilibrium between a gaseous phase and an adsorbed phase in a singlecomponent system. Show that the pressure in the gaseous phase is given by the Langmuir equation \[...

Study smarter with the SolutionInn App


