Consider a system of distinguishable particles with energy levels 0.8.28.38.4&....... For a system with 2 (in...
Fantastic news! We've Found the answer you've been seeking!
Question:
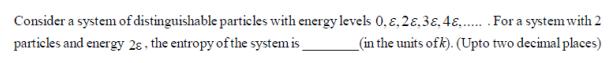
Transcribed Image Text:
Consider a system of distinguishable particles with energy levels 0.8.28.38.4&....... For a system with 2 (in the units ofk). (Upto two decimal places) particles and energy 28, the entropy of the system is_ Consider a system of distinguishable particles with energy levels 0.8.28.38.4&....... For a system with 2 (in the units ofk). (Upto two decimal places) particles and energy 28, the entropy of the system is_
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
A side riser is to be designed for a hollow cylindrical casting of 50 mm inside diameter and 100 mm outside diameter. If the length of cylinder is 50 mm, the diameter of riser in mm is
-
Consider a system of distinguishable particles having only three no degenerate energy levels separated by an energy which is equal to the value of kTat 25.0 K. Calculate (a) The ratio of populations...
-
Consider a system of distinguishable particles having only three energy levels separated by an energy which is equal to the value of KT at 30.0 K. The second energy level is doubly degenerate and the...
-
Which set of parametric equations is shown in the graph below? Explain your reasoning. (a) (b) x = t y = f
-
Nonverbal Communication Around the World Gestures play an important role when people communicate. Because culture shapes the meaning of gestures, miscommunication and misunderstanding can easily...
-
Explain the paradigm mismatch between SQL and programming languages.
-
An auditor is designing an audit program for a computerized payroll and is drafting procedures for payroll preparation, labor cost distribution, and paycheck distribution. Required: For each of the...
-
Heavenly Sounds Corp., an electric guitar retailer, was organized by Mickey Blessing, John Frey, and Nancy Stein. The charter authorized 750,000 shares of common stock with a par of $20. The...
-
You wish to do a series of maintenance plans to automate the maintenance of your SQL server. What tasks should you schedule daily at minimum? List 4
-
In Intuit QuickBooks how do I record this transaction?... Wrote check number 10010 to City Credit Union for payment of amount due (City Credit Union in the Account/Category field); $110.35.
-
Petram Company which is involved in a merchandise business has established a branch at Diredawa with the name -Diredawa Branch. The branch handles sales functions, maintains merchandise inventories,...
-
Based in France and operating in 140 countries, LOral is a global corporate giant. It employs more than 89,000 workers and owns brands such as Maybelline, Ralph Lauren, and Lancme among its...
-
You work on a team with four other members of the customer service team at C-Zone, an auto-parts wholesaler. Your company has around 2,000 employees nationally. The company has low job acceptance...
-
A manufacturing company is preparing to implement a company wellness program within its East Coast, Southeastern, and West Coast locations. These locations employ about 1,800 workers in total. The...
-
Imagine that you are working at a medium-sized business as an HR professional. You are faced with the following dilemmas. Decide how you would handle each issue in the short term and long term. What...
-
It is not unusual for employers to change health insurance plans. For example, an employer may switch to a plan with lower monthly premiums and a higher deductible or may switch from offering a...
-
HP Electronic Ltd can borrow at 6.0%, but currently has no debt. The cost of equity is 12.5%. The current value of the firm is $500,000. What will be the value of equity if HP Electronic borrows...
-
A consultant is beginning work on three projects. The expected profits from these projects are $50,000, $72,000, and $40,000. The associated standard deviations are $10,000, $12,000, and $9,000....
-
Imagine a small electron-sensitive probe of volume 1.00 pm3 inserted into an H+2 molecule-ion in its ground state. Calculate the probability that it will register the presence of an electron at the...
-
A homeowner uses 4.00 x 10J mJ of natural gas in a year to heat a home. Assume that natural gas is all methane, CH4, and that methane is a perfect gas for the conditions of this problem, which are...
-
Explain how a molecule is assigned to a point group.
-
Calculate the volatility and the current price of oil futures implied by the call 1600 August and the call 1700 August of Figure 19.8 by using the Black-Scholes formula with \(T=.25\). OIL CRUDE OIL...
-
Consider a continuous-time environment, with \(e\) as a variable outside of the market. (a) Suppose the final payoff is \(V\left(x_{e}, T ight)=x_{e}(T)\). Find \(V\left(x_{e}, t ight)\). (b) Find a...
-
Mr. Smith wants to buy a car and is deciding between brands A and B. Car A costs \(\$ 20,000\), and Mr. Smith estimates that at the rate he drives he will sell it after 2 years and buy another of the...

Study smarter with the SolutionInn App



