Question: Consider an embedded system which uses Carbon-Zinc D-cell batteries. If the system draws 1.3 mA on average, what is the minimum number of batteries
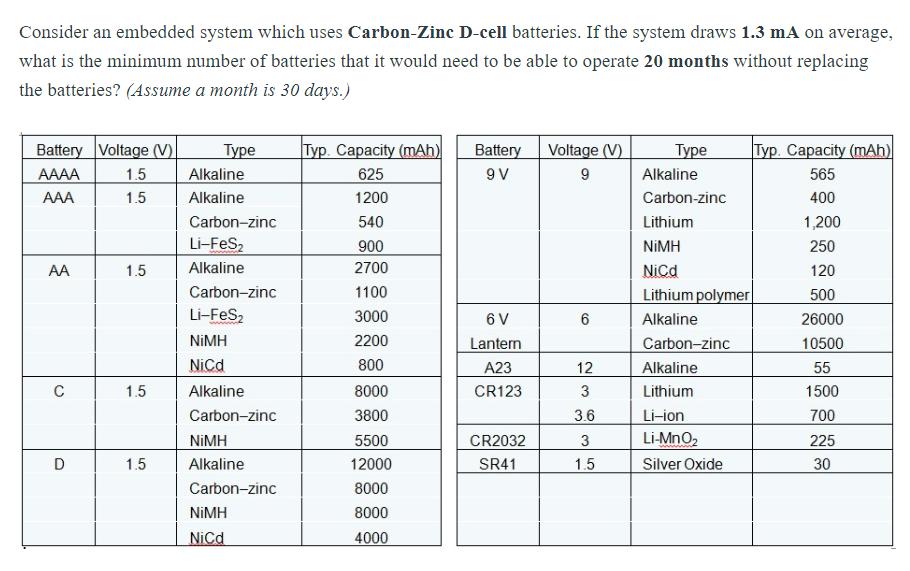
Consider an embedded system which uses Carbon-Zinc D-cell batteries. If the system draws 1.3 mA on average, what is the minimum number of batteries that it would need to be able to operate 20 months without replacing the batteries? (Assume a month is 30 days.) Battery Voltage (V) AAAA 1.5 AAA 1.5 AA C D 1.5 1.5 1.5 Type Alkaline Alkaline Carbon-zinc Li-FeS Alkaline Carbon-zinc Li-FeS NiMH NiCd Alkaline Carbon-zinc NiMH Alkaline Carbon-zinc NiMH NiCd Typ. Capacity (mAh) 625 1200 540 900 2700 1100 3000 2200 800 8000 3800 5500 12000 8000 8000 4000 Battery 9V 6 V Lantern A23 CR123 CR2032 SR41 Voltage (V) 9 6 12 3 3.6 3 1.5 Type Alkaline Carbon-zinc Lithium NiMH NiCd Lithium polymer Alkaline Carbon-zinc Alkaline Lithium Li-ion Li-MnO Silver Oxide Typ. Capacity (mAh) 565 400 1,200 250 120 500 26000 10500 55 1500 700 225 30 Consider an embedded system which uses Carbon-Zinc D-cell batteries. If the system draws 1.3 mA on average, what is the minimum number of batteries that it would need to be able to operate 20 months without replacing the batteries? (Assume a month is 30 days.) Battery Voltage (V) AAAA 1.5 AAA 1.5 AA C D 1.5 1.5 1.5 Type Alkaline Alkaline Carbon-zinc Li-FeS Alkaline Carbon-zinc Li-FeS NiMH NiCd Alkaline Carbon-zinc NiMH Alkaline Carbon-zinc NiMH NiCd Typ. Capacity (mAh) 625 1200 540 900 2700 1100 3000 2200 800 8000 3800 5500 12000 8000 8000 4000 Battery 9V 6 V Lantern A23 CR123 CR2032 SR41 Voltage (V) 9 6 12 3 3.6 3 1.5 Type Alkaline Carbon-zinc Lithium NiMH NiCd Lithium polymer Alkaline Carbon-zinc Alkaline Lithium Li-ion Li-MnO Silver Oxide Typ. Capacity (mAh) 565 400 1,200 250 120 500 26000 10500 55 1500 700 225 30
Step by Step Solution
3.44 Rating (160 Votes )
There are 3 Steps involved in it
To determine the minimum number of batteries needed to operate the embedded system for 20 months wit... View full answer

Get step-by-step solutions from verified subject matter experts


