Consider that the density of states g(E) for super relativistic gas is defined as [1, 0...
Fantastic news! We've Found the answer you've been seeking!
Question:
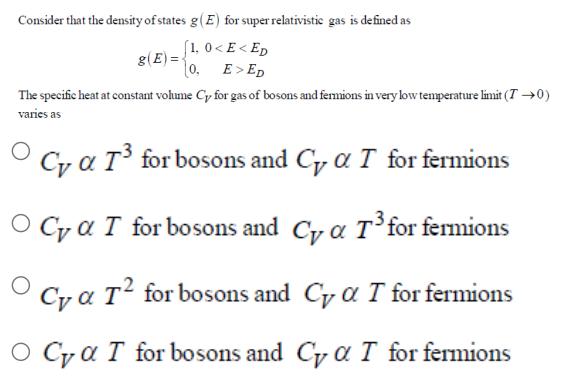
Transcribed Image Text:
Consider that the density of states g(E) for super relativistic gas is defined as [1, 0 ED g(E)= The specific heat at constant volume Cy for gas of bosons and fermions in very low temperature limit (T 0) varies as O Cy a T for bosons and Cy a T for fermions O Ca I for bosons and Cy a T for fermions O Cya T for bosons and C a T for fermions O Ca T for bosons and Cy a T for fermions Consider that the density of states g(E) for super relativistic gas is defined as [1, 0 ED g(E)= The specific heat at constant volume Cy for gas of bosons and fermions in very low temperature limit (T 0) varies as O Cy a T for bosons and Cy a T for fermions O Ca I for bosons and Cy a T for fermions O Cya T for bosons and C a T for fermions O Ca T for bosons and Cy a T for fermions
Expert Answer:
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these physics questions
-
1. A Wigner crystal is a triangular lattice of electrons in a two dimensional plane. The longitudinal vibration modes of this crystal are bosons with dispersion relation w = ak. Show that, at low...
-
For an ideal gas, the specific heat at constant pressure Cp is greater than the specific heat at constant volume Cv because the O A. pressure of the gas remains constant when its temperature remains...
-
A many-body system undergoes a phase transition between two phases A and B at a temperature Te The temperature-dependent specific heat at constant volume Cy of the two phases are given by C) = aT +...
-
Rewrite the following sentences to reflect your audiences viewpoint. 1. We request that you include the order number with your inquiry. 2. We insist that you always bring your credit card to the...
-
Data for Jantzen Company are presented in P12-5A. Instructions Prepare the operating activities section of the statement of cash flows using the direct method.
-
Arun and Margot want to admit Tammy as a third partner for their partnership. Their capital balances prior to Tammys admission are $50,000 each. Prepare a schedule showing how the bonus should be...
-
Using the approach described in Example 2.6, derive the expressions for all the averaged stiffnesses for the planar isotropic lamina in terms of invariants. Use these results to find the...
-
A natural gas containing 95 mole% methane and the balance ethane is burned with 20.0% excess air. The stack gas which contains no unburned hydrocarbons or carbon monoxide leaves the furnace at 900?C...
-
Zack is using the Treynor and Black optimisation method. He has determined that the optimal weighting in the active portfolio is 76.19%. The active portfolio has a residual standard deviation of 5%...
-
Erica and Bob participate in a friendly Hackathon that allows each to solve one question a day out of the three offered. There will be one easy, one medium and one hard question, with points awarded...
-
Let T: P(R) R2 and T(a + bt) -> ["+"]. Find T-1
-
Beverly Bergman purchased land for $30,000 in 2009. The land was valued at $180,000 on April 15, 2019, when Beverly died. Her son Jack inherited the land. Six months later, on October 15, 2019, the...
-
Brian owns 200 shares of Bell Corp. stock purchased in January 2017 for $2,400. On January 30, 2019, he receives 200 taxable stock rights valued at $5 with the right to purchase additional shares at...
-
Solve simple linear programming problems using the graphical method.
-
Spend some time studying Yammer (www.yammer.com), which is an enterprise social network. What is the basis of Yammers business opportunity? Evaluate Yammer on the four essential qualities of an...
-
Sarah Shore bought 500 shares of stock on October 22, 2018, for $30,000. On March 25, 2019, she gave half the shares to her sister Cindy when the fair market value of the shares was $55 per share. If...
-
A six year project requires an increase in inventories of 100,000$ at the start of the project that will no longer be needed at the end of the project. If the opportunity cost of capital is 5%. What...
-
Use the following data to answer the next two (2) questions: Product 1 Product 2 Product 3 Direct Material Cost $25,000 $30,000 $35,000 Direct Labor Cost $30,000 $40,000 $50,000 Direct Labor Hours...
-
The Helmholtz function of a substance has the form
-
An adiabatic heat exchanger is used to heat cold water at 15C entering at a rate of 5 kg/s by hot water at 90C entering at a rate of 4 kg/s. If the exit temperature of hot water is 50C, the exit...
-
A wind turbine with a blade diameter of 25 m is to be installed in a location where average wind velocity is 6 m/s. If the overall efficiency of the turbine is 34 percent, determine (a) The average...
-
How much heat is to be added to convert \(4 \mathrm{~kg}\) of water at \(20^{\circ} \mathrm{C}\) in to steam at 8 bar and \(200^{\circ} \mathrm{C}\). Take \(C_{p}\) of superheated steam as \(2.1...
-
What do you understand by mechanical and thermal efficiency? A steam plant uses 3 tonne of \(\mathrm{coal} / \mathrm{h}\). The steam is fed to turbine the output of which is \(4 \mathrm{MW}\). The...
-
Determine the total heat content per unit mass at the following state using the steam tables. Assume ambient pressure to be \(100 \mathrm{kPa}\) and \(C_{P}\) \(=2.0934 \mathrm{~kJ} / \mathrm{kg}\)....

Study smarter with the SolutionInn App



