Consider the ammonia process given below. A feed of 20% N2, 78% H2 and 2% CH4 is
Fantastic news! We've Found the answer you've been seeking!
Question:
Consider the ammonia process given below. A feed of 20% N2, 78% H2 and 2% CH4 is mixed with two recycle streams and enters a reactor. Here, conversion per pass of N2 to NH3 is 45% according to the reaction:
N2 + 3H2 --> 2NH3
The ammonia product is recovered by flashing the reactor effluent in two stages and recycling the overhead vapour. If the purge fraction is 5% for the high pressure recycle, what is the methane concentration in the reactor feed?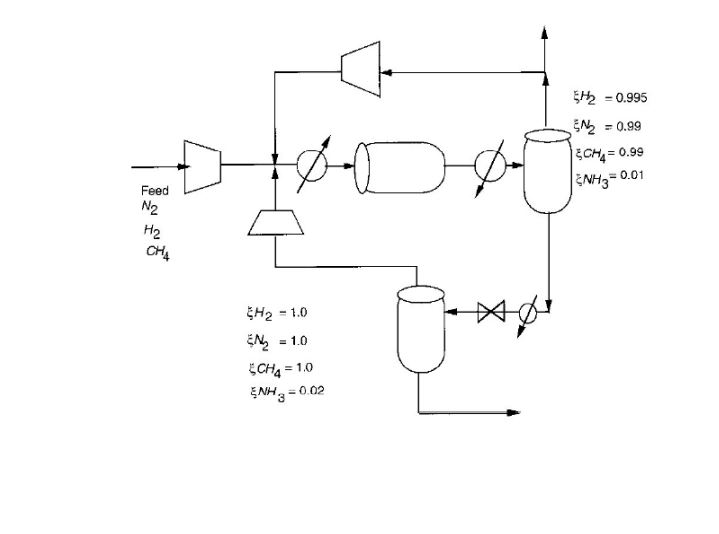
Related Book For 

Posted Date:




