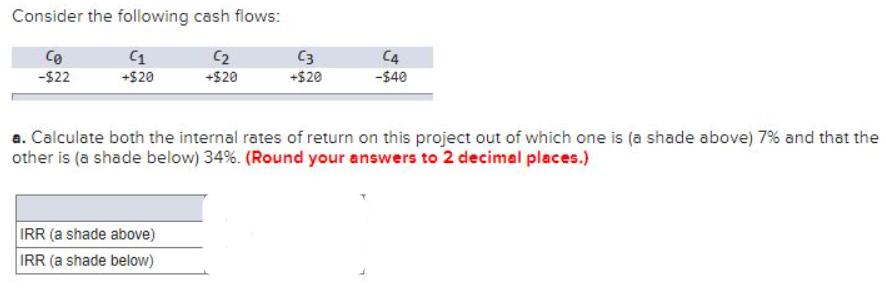
Consider the following cash flows: (1 Co -$22 +$20 C +$20 IRR (a shade above) IRR...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the following cash flows: (1 Co -$22 +$20 C₂ +$20 IRR (a shade above) IRR (a shade below) C3 +$20 C4 -$40 a. Calculate both the internal rates of return on this project out of which one is (a shade above) 7% and that the other is (a shade below) 34%. (Round your answers to 2 decimal places.) Consider the following cash flows: (1 Co -$22 +$20 C₂ +$20 IRR (a shade above) IRR (a shade below) C3 +$20 C4 -$40 a. Calculate both the internal rates of return on this project out of which one is (a shade above) 7% and that the other is (a shade below) 34%. (Round your answers to 2 decimal places.)
Expert Answer:
Answer rating: 100% (QA)
The internal rate of return IRR is the discount rate that makes the net present value NPV of a series of cash flows equal to zero The cash flows from ... View the full answer

Related Book For 

Corporate Finance
ISBN: 978-0077861759
10th edition
Authors: Stephen Ross, Randolph Westerfield, Jeffrey Jaffe
Posted Date:
Students also viewed these finance questions
-
The risk hee rate is 1 99% and the market risk premium is 5 98% A stock whap of 96 just paid a dividend of $123 The dividend is expected to grow at 21.37% for five years and then grow at 4 75%...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Consider the following cash flows on two mutually exclusive projects for the Bahamas Recreation Corporation (BRC). Both projects require an annual return of 14 percent. As a financial analyst for...
-
The dehydration butanol of alumina is carried out over a silica-alumina catalyst at 680K. CH3CH2CH2CH20H------->cat CH3CH=CHCH3 + H2O The rate law is -r Bu = KPBU/(1+KBuPBul with k= 0.054...
-
1. Why must a film used to observe interference colors be thin? 2. Suppose that the central diffraction maximum for two slits contains 17 interference fringes for some wavelength of light. How many...
-
A sailboat costs $35,000. You pay 20% down and amortize the rest with equal monthly payments over a 12-year period. If you must pay 8.75% compounded monthly, what is your monthly payment? How much...
-
What steps should be taken by the auditor after he has received a cutoff statement?
-
Keiffer Production manufactures three joint products in a single process. The following information is available for August 2010: Allocate the joint cost of $558,000 to the production based on the a....
-
Surgery Center is an outpatient surgical clinic that was profitable for many years, but Medicare has cut its reimbursements by as much as 30% As a result, the clinic wants to better understand its...
-
Darcelle teaches skiing at the Snow School. Jack runs a ski shop called Up Hill, Down Hill. Their combined bank interest is $1,000. If they get a refund, they would like the money deposited into...
-
Bottlebot, Inc. an S Corporation had the following asset sales over the past three years. In year one, the company sold a car, purchase price $40,000, the company took $15,000 of depreciation and...
-
Victor Korchnoi bought a bond one month before a semi-annual coupon was due. The face value was $10,000 and the coupon rate 8.5%. At the time of purchase there were 34 coupons left and the YTM was 6%...
-
Sphere Foods Company produces a candy called "Little Stars," a bag of dozen, individually wrapped, star-shaped candies made primarily from a blend chocolate, macadamia nuts, and a blend of heavy...
-
25. If a current of 0.40 A is used to electrolyze a 0.50 M solution of Au3+(aq) for 35.0 minutes, how much Au(s) will be electroplated onto to a ring immersed in this solution? (Assume 100% current...
-
What is the pH of a 0.253M solution of a monoprotic weak acid with a Ka of 4.9x10^-6. You can use the 5% approx rule.
-
A gas centrifuge enrichment plant operates at negative 30C. The inner radii measure 10.0 cm and have a linear velocity of 450 m/s. Determine the separation factor of this centrifuge. Plot the number...
-
Max Weber's - Life Chances Goal: Compare facts and data about styles of life and standards of living in the different types of poverty worldwide.To their own standard of living and norms in their...
-
What mass of H2 will be produced when 122 g of Zn are reacted? Zn(s) + 2HCl(aq) ( ZnCl2(aq) + H2(g)
-
In practice, the use of the dividend discount model is refined from the method we presented in the textbook. Many analysts will estimate the dividend for the next five years and then estimate a...
-
In May 2004, Sysco Corporation, the distributor of food and food-related products (not to be confused with Cisco Systems), announced it had signed an interest rate swap. The interest rate swap...
-
National Electric Company (NEC) is considering a $45 million project in its power systems division. Tom Edison, the companys chief financial officer, has evaluated the project and determined that the...
-
How does attribution theory allow managers to justify workers behaviors?
-
What are common methods to measure a persons attitude?
-
Is stereotyping negative or positive? Why?

Study smarter with the SolutionInn App


