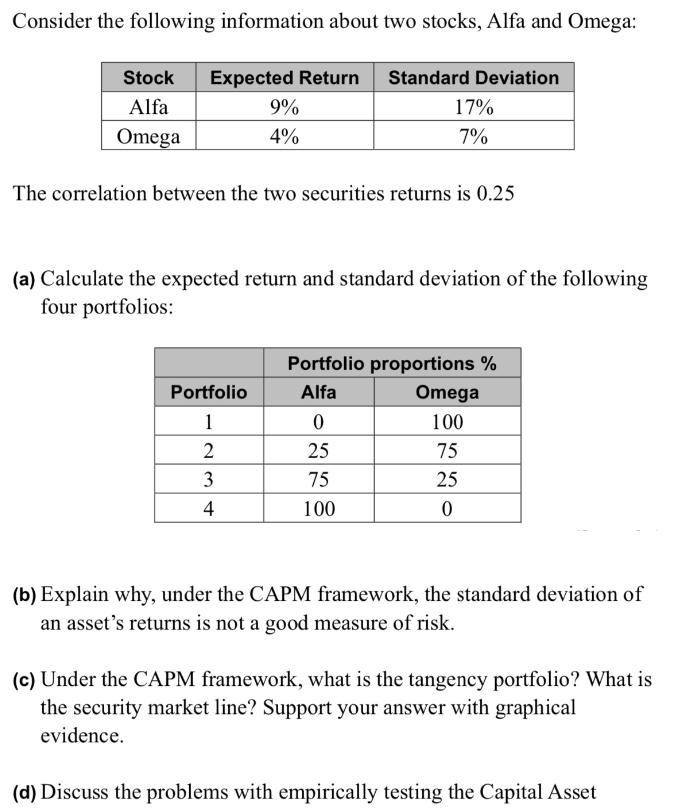
Consider the following information about two stocks, Alfa and Omega: Stock Expected Return Standard Deviation Alfa...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the following information about two stocks, Alfa and Omega: Stock Expected Return Standard Deviation Alfa Omega The correlation between the two securities returns is 0.25 Portfolio 1 9% 4% (a) Calculate the expected return and standard deviation of the following four portfolios: 234 17% 7% 4 Portfolio proportions % Alfa 0 25 75 100 Omega 100 75 25 0 (b) Explain why, under the CAPM framework, the standard deviation of an asset's returns is not a good measure of risk. (c) Under the CAPM framework, what is the tangency portfolio? What is the security market line? Support your answer with graphical evidence. (d) Discuss the problems with empirically testing the Capital Asset Consider the following information about two stocks, Alfa and Omega: Stock Expected Return Standard Deviation Alfa Omega The correlation between the two securities returns is 0.25 Portfolio 1 9% 4% (a) Calculate the expected return and standard deviation of the following four portfolios: 234 17% 7% 4 Portfolio proportions % Alfa 0 25 75 100 Omega 100 75 25 0 (b) Explain why, under the CAPM framework, the standard deviation of an asset's returns is not a good measure of risk. (c) Under the CAPM framework, what is the tangency portfolio? What is the security market line? Support your answer with graphical evidence. (d) Discuss the problems with empirically testing the Capital Asset
Expert Answer:
Answer rating: 100% (QA)
a Calculate the expected return and standard deviation of the following four portfolios Portfolio Pr... View the full answer

Related Book For 

Fundamentals of Financial Management
ISBN: 978-1305635937
Concise 9th Edition
Authors: Eugene F. Brigham
Posted Date:
Students also viewed these finance questions
-
Consider the following information about two stocks (D and E) and two common risk factors (1 and 2): a. Assuming that the risk-free rate is 5.0%, calculate the levels of the factor risk premia that...
-
There are three assets, A, B and C, where A is the market portfolio and C is the risk-free asset. The return on the market has a mean of 12% and a standard deviation of 20%. The risk-free asset...
-
What volume of 0.2 M AGNO3 is required to react exactly with 120.0 mL of 0.3 M Na SO4 solution? Hint: You will want to write a balanced reaction. 180mL 90mL O345 ml 360mL O 36mL
-
Please help me calculate the current assets and current liabilities. Cash and cash equivalents Deposits Marketable securities Inventory Property & equipment, net Loan to shareholders Notes receivable...
-
Analyzing and Interpreting the Effects of Inventory Errors The income statement for Pruitt Company summarized for a four-year period shows the following: An audit revealed that in determining these...
-
Change the given quantity to the quantity indicated. 0.52 km to decimeters.
-
In a reaction, if \(H\) and \(S\) are positive, the rate of reaction (a) Increases with increase in temperature (b) Decreases with increase in temperature (c) Has no effect on temperature (d)...
-
OBrien Company manufactures and sells one product. The following information pertains to each of the companys first three years of operations: Variable costs per unit: Manufacturing: Direct materials...
-
Lopez Company is considering replacing one of its old manufacturing machines. The old machine has a book value of $45,000 and a remaining useful life of five years. It can be sold now for $52,000....
-
Rainmaker Environmental Consultants is just finishing its second year of operations. The companys unadjusted trial balance at October 31, 2023, follows. Rainmaker prepares adjustments each October...
-
The maximum speed of a truck up a steep hill is 50mph with no cargo. With 20 tons of cargo, its maximum speed is 40mph. Calculate the rate of change and explain the meaning of this rate of change in...
-
Complete a frequency distribution for the following data that represents the recent sales prices (in $1,000s) of homes in Midwestern County. As this is quantitative data, calculate five classes,...
-
As part of a weight reduction program, a man designs a monthly exercise program consisting of bicycling, jogging, and swimming. He would like to exercise at most 40 hours, devote at most 8 hours to...
-
Find the third derivative of the function f(x)=x-3x4
-
An accounting firm has just contacted your consulting business for assistance. They want to hire someone to do accounting and payroll. The bookstore is downstairs, while the office is upstairs in an...
-
As in Quiz10, assume we have this database: CREATE TABLE 'country' ( 'Code' CHAR (3) NOT NULL DEFAULT" 'Name' CHAR (52) NOT NULL DEFAULT", CECS 535: Introduction to Database Fall 2023, Homework 5...
-
Besides an introduction to operations in your reading, you read about different quality control strategies and practice with some of the tools to ensure high-quality products/services. Based on your...
-
In a large midwestern university, 30% of the students live in apartments. If 200 students are randomly selected, find the probability that the number of them living in apartments will be between 55...
-
The real risk-free rate, r* , is 1.7%. Inflation is expected to average 1.5% a year for the next 4 years, after which time inflation is expected to average 4.8% a year. Assume that there is no...
-
a. Set up an amortization schedule for a $19,000 loan to be repaid in equal installments at the end of each of the next 3 years. The interest rate is 8% compounded annually. b. What percentage of the...
-
For 2016, Gourmet Kitchen Products reported $22 million of sales and $19 million of operating costs (including depreciation). The company has $15 million of total invested capital. Its after-tax cost...
-
Justify the following statement with illustration: 'Violation of Kelvin-Planck statement leads to the violation of Clausius statement'.
-
(a) Two iron blocks of same size and at distinct temperatures \(T_{1}\) and \(T_{2}\) are brought in thermal contact with each other. The transfer process is allowed to take place until the thermal...
-
A spherical balloon of \(1 \mathrm{~m}\) diameter contains a gas at \(120 \mathrm{kPa}\). The gas inside the balloon is heated until the pressure reaches \(360 \mathrm{kPa}\). During heating the...

Study smarter with the SolutionInn App


