Consider the following reaction: 8NH3(g) + 3Cl2(g) ----> 6NH4Cl(s) + N(g) What is the volume of...
Fantastic news! We've Found the answer you've been seeking!
Question:
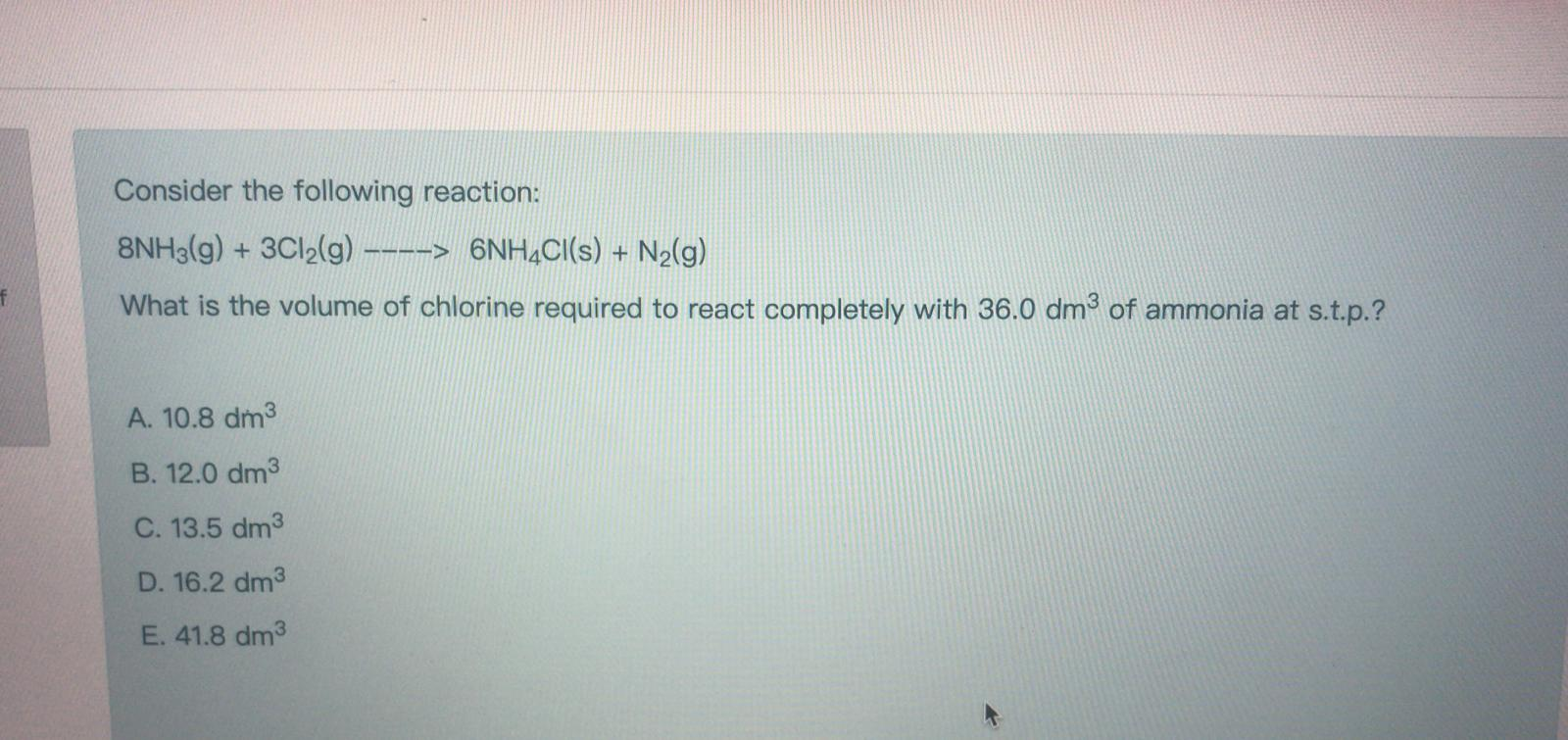
Transcribed Image Text:
Consider the following reaction: 8NH3(g) + 3Cl2(g) ----> 6NH4Cl(s) + N₂(g) What is the volume of chlorine required to react completely with 36.0 dm³ of ammonia at s.t.p.? A. 10.8 dm³ B. 12.0 dm³ C. 13.5 dm³ D. 16.2 dm³ E. 41.8 dm³ Consider the following reaction: 8NH3(g) + 3Cl2(g) ----> 6NH4Cl(s) + N₂(g) What is the volume of chlorine required to react completely with 36.0 dm³ of ammonia at s.t.p.? A. 10.8 dm³ B. 12.0 dm³ C. 13.5 dm³ D. 16.2 dm³ E. 41.8 dm³
Expert Answer:
Answer rating: 100% (QA)
8NH3 3C1 6NH4Cl N 3 mals chlarine rea for 8 malls NH3 for ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction at 800. K: N2(g) + 3F2(g) 2NF3(g) An equilibrium mixture contains the following partial pressures: PN2 = 0.021 atm, PF2 = 0.063 atm, and PNF3 = 0.48 atm. Calculate Go...
-
Consider the following reaction at 298 K: 2SO2(g) + O2(g) 2SO3(g) An equilibrium mixture contains O2(g) and SO3(g) at partial pressures of 0.50 atm and 2.0 atm, respectively. Using data from...
-
Product Strategy Duff has always followed a level production strategy because Duff prefers not to fire and hire employees. Management is open to change if there is enough of a cost benefit for the...
-
1. Consumer surplus equals _________ minus _________. 2. Producer surplus equals _________ minus_________. 3. In Figure Tupaks consumer surplus is _________, compared to _________ for Thurl. 4. In...
-
Microprocessor X has 30 instructions in its instruction set, and microprocessor Y has 45 instructions in its instruction set. You are told that Y is a RISC processor. Can you conclusively say that X...
-
Elite Mobile Homes reported the following in its financial statements for the year ended December 31, 2007 (adapted, in millions): .Determine the following for Elite Mobile Homes during 2007: a....
-
Buckeye Department Stores, Inc., operates a chain of department stores in Ohio. The companys organization chart appears below. Operating data for 20x5 follow. The following fixed expenses are...
-
Recently a large sales company computerised their sales operation to improve their profit and speed up their services. This company has many branches at different locations. The company introduced...
-
Imagine that you are Magna's new corporate controller and answer the following: 1. Describe Magna's strategy in terms of how it competes for customers. 2. Based on Magna's strategy and the data...
-
Navajo Company's financial statements show the following. The company recently discovered that in making physical counts of y is inventory, it had made the following errors. Year 1 ending inventory...
-
Toby and Norma are married at 18 and quickly have three children. Eventually, Toby and Norma separate and their children grow into adults and have their own families. Norman ever remarries and passes...
-
Prior news related to COVID supply bottlenecks, I really thought the overall "SCM" concept was still relatively new to many organizations. Previously, we (in my former apparel company) had...
-
A mass (m = 4.31kg) is connected to a spring (k = 182.4N/m) on the surface of a horizontal, frictionless table and constrained to move in the j direction (gravity is pointed in the k direction). When...
-
The year ended 3 0 June 2 0 2 3 : 1 . Contributed equity on 1 July 2 0 2 2 consisted of 1 million ordinary shares fully paid to $ 2 each . Other equity account balances on 1 July 2 0 2 2 were:...
-
On 1 5 April 2 0 2 1 , Growth Realty Ltd purchased a piece of equipment worth $ 2 0 0 , 0 0 0 by accepting a 6 - month discounted bank bill. The bill has a face value of $ 2 0 9 , 0 0 0 and is due...
-
Jimmy's Painting BUS 323 Fall 2023 Group Project Case Jimmy's Painting is an owner-operator run sole proprietorship that operates in Galloway, BC. Jimmy Davis started the company in several years...
-
As you rewrite these sentences, replace the cliches and buzzwords with plain language (if you don't recognize any of these terms, you can find definitions online): a. Being a jack-of-all-trades, Dave...
-
Draw all enol isomers of the following compounds. If there are none, explain why. (a) 2-methylpentanoic acid (b) Benzaldehyde
-
Give the products (if any) expected from the treatment of each of the following compounds with ozone followed by dimethyl sulfide (a) (b) 2-methylpentane CH2
-
Within each set, which compound should show NMR absorptions with the greater chemical shifts? Explain your choices. (1) (2)
-
Under what conditions can WTO members not use MFN when dealing with one another?
-
What does most favored nation (MFN) mean?
-
How do the various forms of economic integration differ?

Study smarter with the SolutionInn App


