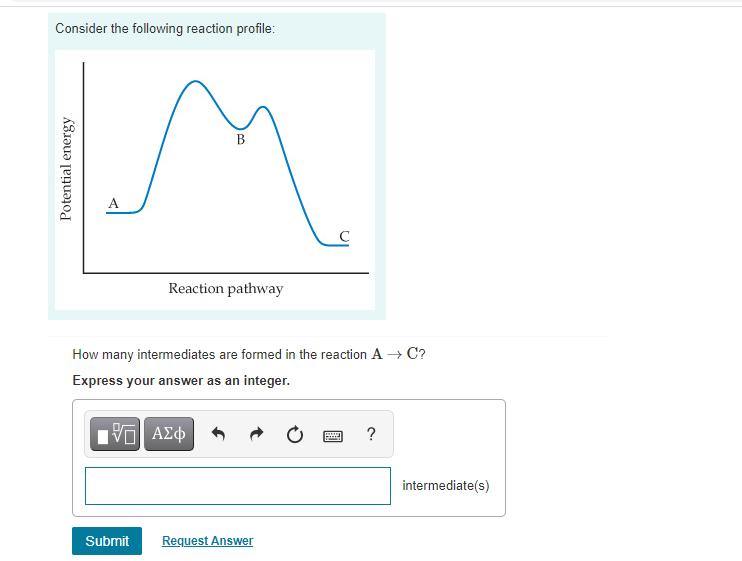
Consider the following reaction profile: Potential energy B Reaction pathway How many intermediates are formed in...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
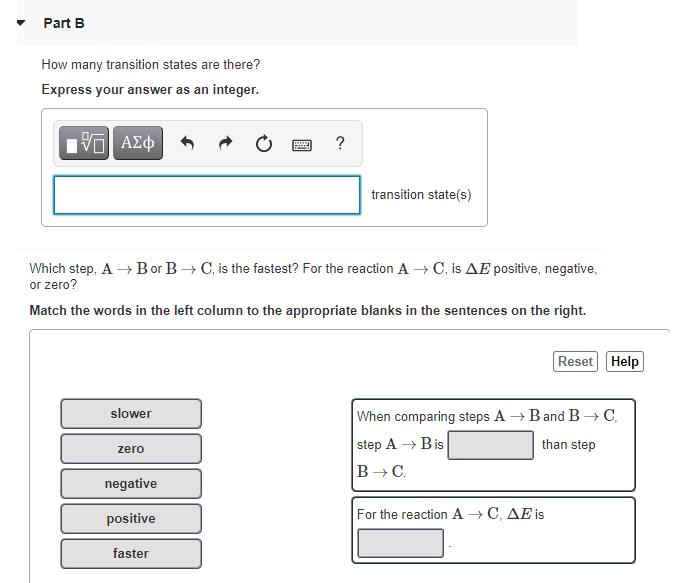
Consider the following reaction profile: Potential energy B Reaction pathway How many intermediates are formed in the reaction A→ C? Express your answer as an integer. IVE ΑΣΦ Submit Request Answer F ? intermediate(s) Part B How many transition states are there? Express your answer as an integer. 197| ΑΣΦ slower Which step, A → Bor B → C, is the fastest? For the reaction A→ C, is AE positive, negative, or zero? Match the words in the left column to the appropriate blanks in the sentences on the right. zero ? negative positive faster transition state(s) Reset Help When comparing steps A→ B and B → C, than step step A → Bis B → C. For the reaction A → C, AE is All is one question Consider the following reaction profile: Potential energy B Reaction pathway How many intermediates are formed in the reaction A→ C? Express your answer as an integer. IVE ΑΣΦ Submit Request Answer F ? intermediate(s) Part B How many transition states are there? Express your answer as an integer. 197| ΑΣΦ slower Which step, A → Bor B → C, is the fastest? For the reaction A→ C, is AE positive, negative, or zero? Match the words in the left column to the appropriate blanks in the sentences on the right. zero ? negative positive faster transition state(s) Reset Help When comparing steps A→ B and B → C, than step step A → Bis B → C. For the reaction A → C, AE is All is one question
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction at 800. K: N2(g) + 3F2(g) 2NF3(g) An equilibrium mixture contains the following partial pressures: PN2 = 0.021 atm, PF2 = 0.063 atm, and PNF3 = 0.48 atm. Calculate Go...
-
Consider the following reaction at 298 K: 2SO2(g) + O2(g) 2SO3(g) An equilibrium mixture contains O2(g) and SO3(g) at partial pressures of 0.50 atm and 2.0 atm, respectively. Using data from...
-
In a recent survey, 80% of the community favored building a police substation in their neighborhood. If 20 citizens are chosen, what is the mean and standard deviation for the number favoring the...
-
Find the binding energy of a 147N nucleus in MeV if the mass of the neutral N atom is 14.00307 u
-
Which of the following sequences of sentences makes this paragraph most logical? F. NO CHANGE G. Who is important to you H. Who means a lot to you J. OMIT the underlined portion.
-
How can compensation design help mitigate agency problems?
-
An aging analysis on June 30 of the accounts receivable of U-Z Door Corporation indicates that uncollectible accounts amount to $86,000. Prepare the journal entry to record uncollectible accounts...
-
(a) The Treasury desk of a global bank incorporated in UK wants to invest GBP 200 million on 1st January, 2019 for a period of 6 months and has the following options: (1) The Equity Trading desk in...
-
This exercise concerns TM M 1 , whose description and state diagram appear in Example 3.9. In each of the parts, give the sequence of configurations that M1 enters when started on the indicated input...
-
Vaughn Manufacturing had 829000 shares of common stock issued and outstanding at December 31, 2017. On July 1, 2018, an additional 41600 shares of common stock were issued for cash. Vaughn also had...
-
Reconsider question 57, but this time assume that each population has three members. Question 57 Suppose you have three populations containing two members each. Suppose the means and the variances of...
-
Suppose a bag contains 12 balls distributed as follows: five red dotted balls, two red striped balls, one gray dotted ball, and four gray striped balls. (a) Suppose you draw a red ball from the bag....
-
Find the probability of rolling a 1 on the first roll of a die, a 2 on the second, and a 3 on the third.
-
Find the probability of a 5 turning up at least once in 2 tosses of a fair die.
-
Country C can produce 200 tons of wheat or 50 million automobiles per year. Country D can produce 500 tons of wheat or 125 million automobiles per year. The production possibilities curve for each...
-
4. [2+3=5 points] Evaluate the following limits. No points will be given if L'Hopital's rule is used: (a) lim x6 2x+12 x + 6 2x x6 2 + x (b) lim
-
The comparative statements of financial position of Menachem NV at the beginning and end of the year 2019 appear below. Net income of ¬34,000 was reported, and dividends of ¬23,000 were paid...
-
Write the condensed structural formula for each of the following compounds: (a) 2-ethyl-1-hexanol, (b) methyl phenyl ketone, (c) para-bromobenzoic acid, (d) ethyl butyl ether, (e) N,...
-
(a) What is the meaning of the standard free-energy change, G, as compared with G? (b) For any process that occurs at constant temperature and pressure, what is the significance of G = 0? (c) For a...
-
(a) What is an ideal solution? (b) The vapor pressure of pure water at is 60 oC is 149 torr. The vapor pressure of water over a solution at 60 oC containing equal numbers of moles of water and...
-
a. Write out and explain the equation for volume breakeven. b. What is the difference between accounting breakeven and economic breakeven?
-
What is marginal analysis?
-
a. What is cost shifting? b. What is cross-subsidization?

Study smarter with the SolutionInn App



