Consider the following reaction:2CH3OH(g)+3O2(g)?2CO2(g)+4H2O(g) Each of the following moleculardiagrams represents an initial mixture of the reactants. (Figure
Fantastic news! We've Found the answer you've been seeking!
Question:
Consider the following reaction:2CH3OH(g)+3O2(g)?2CO2(g)+4H2O(g) Each of the following moleculardiagrams represents an initial mixture of the reactants. (Figure 1)How many CO2 molecules would be formed by complete reaction in eachcase? (Assume 100% actual yield.)
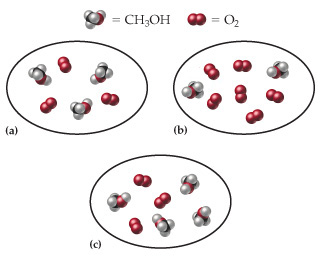
a)Express your answer as an integer
b)Express your answer as an integer
c)Express your answer as aninteger.
Related Book For 

Posted Date:




