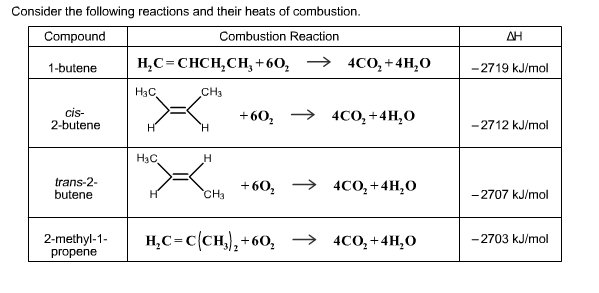
Consider the following reactions and their heats of combustion. Compound Combustion Reaction 1-butene HC=CHCHCH +604CO+ 4HO...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Consider the following reactions and their heats of combustion. Compound Combustion Reaction 1-butene H₂C=CHCH₂CH₂ +60₂4CO₂+ 4H₂O CH3 cis- 2-butene trans-2- butene 2-methyl-1- propene H3C, H H₂C H H H CH3 +60₂ → 4CO₂ + 4H₂O +60₂ → 4CO₂+ 4H₂O H₂C=C(CH₂)₂+60₂ → 4CO₂+ 4H₂O ΔΗ -2719 kJ/mol -2712 kJ/mol -2707 kJ/mol -2703 kJ/mol With the above information and your knowledge of alkenes, select the statements that are TRUE. 1-Butene is the most stable compound. 2-Methyl-1-propene is the most stable compound. 1-Butene is the least stable compund. 2-Methyl-1-propene is the least stable compound. The more carbon atoms attached to the double bond, the more stable the alkene. A trans isomer is less stable than a cis isomer due to more steric hindrance. sp² hybridized carbon atoms are more electronegative than sp³ hybridized atoms. Consider the following reactions and their heats of combustion. Compound Combustion Reaction 1-butene H₂C=CHCH₂CH₂ +60₂4CO₂+ 4H₂O CH3 cis- 2-butene trans-2- butene 2-methyl-1- propene H3C, H H₂C H H H CH3 +60₂ → 4CO₂ + 4H₂O +60₂ → 4CO₂+ 4H₂O H₂C=C(CH₂)₂+60₂ → 4CO₂+ 4H₂O ΔΗ -2719 kJ/mol -2712 kJ/mol -2707 kJ/mol -2703 kJ/mol With the above information and your knowledge of alkenes, select the statements that are TRUE. 1-Butene is the most stable compound. 2-Methyl-1-propene is the most stable compound. 1-Butene is the least stable compund. 2-Methyl-1-propene is the least stable compound. The more carbon atoms attached to the double bond, the more stable the alkene. A trans isomer is less stable than a cis isomer due to more steric hindrance. sp² hybridized carbon atoms are more electronegative than sp³ hybridized atoms.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Consider the following reactions at some temperature: 2NOCl( g) 2NO( g) + Cl2( g) K = 1.6 1025 2NO( g) N2( g) + O2( g) K = 1 1031 For each reaction some quantities of the reactants were placed in...
-
Given the following reactions and their enthalpies: a. Devise a way to calculate H for the reaction H2O(g) 2H(g) + O(g) b. From this, estimate the H - O bond energy. (kJ/mol) +436 +495 H2(g)- 2H(g)...
-
Give the products of the following reactions and their configurations: HBr peroxide Br2, CH2CI2 CH3 0 Br2, H2o
-
Alleghany Community College operates four departments. The square footage used by each department is shown below. Alleghany's annual building rental cost is $320,000 What amount of rent expense that...
-
The following information appears in a footnote to the 2012 financial statements of Nike, Inc.: Requirements 1. Based on this information, what do you think are Nike, Inc.s reportable segments as of...
-
What are the advantages and disadvantages for both employers and employees of resolving disputes through ADR? Why would a company have an employee sign a commitment to resolve disputes through ADR?
-
Quilts R Us (QRU) is considering investing in a new patterning attachment with the cash flow profile shown in the table below. QRU's MARR is 13.5 percent/year. a. What is this investment's external...
-
On May 1, 2017, Herron Corp. issued $600,000, 9%, 5-year bonds at face value. The bonds were dated May 1, 2017 and pay interest annually on May 1. Financial statements are prepared annually on...
-
Develop an algorithm, in the form of a Raptor flowchart, to compute and display the amount of federal income tax to be paid when provided with the income as input. The Canadian Federal income tax...
-
The neural structure of the sea slug Aplysia has been widely studied (first by Nobel Laureate Eric Kandel) because it has only about 20,000 neurons, most of them large and easily manipulated....
-
Calculate the total mortgage cost for one year for bi-weekly mortgage payments of $654.22. $34 019.44 $7850.64 $17 009.72 $15 701.28 Question 6 (1 point) Calculate the property tax payment for $3578...
-
Today we are going to play the "Questionizer" game. I will start by asking a question related to Lesson 1. The first student to respond to my question will do his or her best to answer it and then...
-
Read the given case study and answer the following questions (Question 1-Question 5) You are appointed by a local consulting company for a project to design and implement an employee's records...
-
Scenario one Each regional site will produce for their own region Demand is met! No site has exceeded their capacity! Minimized fixed costs by running each site at 200k Total cost (fixed and...
-
To find the utility-maximizing combination of goods x and y for this consumer, we can use the concept of consumer optimization subject to a budget constraint. The consumer's goal is to maximize...
-
Unemployment insurance benefits help individuals who have lost their job to sustain a desirable consumption level. An MIT economist, Jonathan Gruber, argues that private insurance or savings are not...
-
Explain two ways in which the trachea is adapted to perform its functions.
-
Read Case Study Google: Dont Be Evil Unless and answer the following: Why do you think Google was adamant about not wanting to supply information requested by the government concerning the Child...
-
Molybdenum is obtained as a by-product of copper mining or is mined directly (primary deposits are in the Rocky Mountains in Colorado). In both cases it is obtained as MoS2, which is then converted...
-
A spherical glass container of unknown volume contains helium gas at 25 o C and 1.960 atm. When a portion of the helium is withdrawn and adjusted to 1.00 atm at 25 o C, it is found to have a volume...
-
The melting point of a fictional substance X is 225oC at 10.0 atm. If the density of the solid phase of X is 2.67 g/cm3 and the density of the liquid phase is 2.78 g/cm3 at 10.0 atm, predict whether...
-
Find P 80 , the 80th percentile for the red blood cell counts of women.
-
Mensa International calls itself the international high IQ society, and it has more than 100,000 members. Mensa states that candidates for membership of Mensa must achieve a score at or above the...
-
A new integrated circuit board is being developed for use in computers. In the early stages of development, a lack of quality control results in a 0.2 probability that a manufactured integrated...

Study smarter with the SolutionInn App


