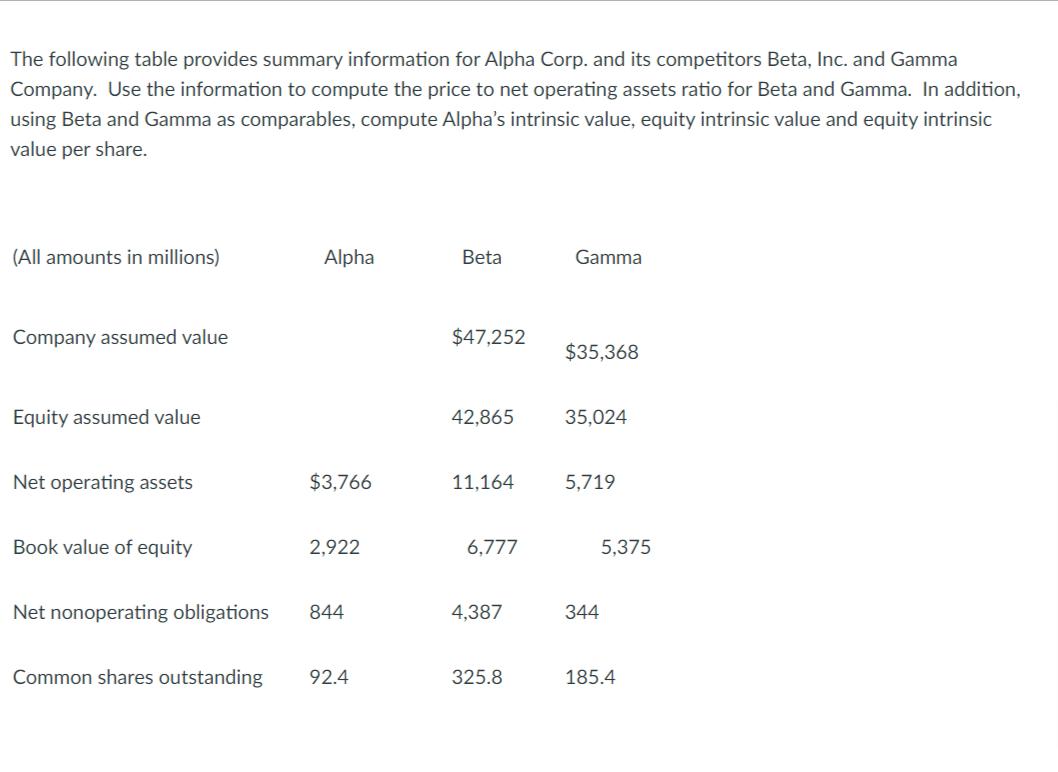
The following table provides summary information for Alpha Corp. and its competitors Beta, Inc. and Gamma...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The following table provides summary information for Alpha Corp. and its competitors Beta, Inc. and Gamma Company. Use the information to compute the price to net operating assets ratio for Beta and Gamma. In addition, using Beta and Gamma as comparables, compute Alpha's intrinsic value, equity intrinsic value and equity intrinsic value per share. (All amounts in millions) Company assumed value Equity assumed value Net operating assets Book value of equity Net nonoperating obligations Alpha $3,766 2,922 844 Common shares outstanding 92.4 Beta $47,252 42,865 11,164 6,777 4,387 325.8 Gamma $35,368 35,024 5,719 344 5.375 185.4 The following table provides summary information for Alpha Corp. and its competitors Beta, Inc. and Gamma Company. Use the information to compute the price to net operating assets ratio for Beta and Gamma. In addition, using Beta and Gamma as comparables, compute Alpha's intrinsic value, equity intrinsic value and equity intrinsic value per share. (All amounts in millions) Company assumed value Equity assumed value Net operating assets Book value of equity Net nonoperating obligations Alpha $3,766 2,922 844 Common shares outstanding 92.4 Beta $47,252 42,865 11,164 6,777 4,387 325.8 Gamma $35,368 35,024 5,719 344 5.375 185.4
Expert Answer:
Answer rating: 100% (QA)
Solution ACalculation of price to net operating assets ratio Price to net oper... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A spark ignition engine based on Otto cycle has a working temperature range of 25.0 C and 700.0 C and its output power is 200 kW. The efficiency of this engine is found to be 56.5%. a. Calculate the...
-
Consider the following statement in predicate calculus: empty_class(C) X[takes(X; C)] (a) Translate this statement to clausal form. (b) Can you translate the statement into Prolog? Does it make a...
-
Q.1 Consider a typical consumer with a monthly income of Rs. 240. His consumption bundle is composed of only two commodities: cola (x) and fries (y). The market prices for each unit of these...
-
In recent years, Avery Transportation purchased three used buses. Because of frequent turnover in the accounting department, a different accountant was in charge of selecting the depreciation method...
-
A paired sample study has been conducted to determine whether two populations have equal means. Twenty paired samples were obtained with the following sample results: Based on these sample data and a...
-
An extruder has a barrel diameter of 100 mm. The screw rotates at 100 rpm, has a channel depth of 6 mm, and a flight angle of 17.5. What is the highest flow rate of polypropylene that can be achieved?
-
Assume the following exceptions to prescribed control procedures over payroll transactions occurred in the Haze Company: 1. A fictitious employee is on payroll. 2. A fictitious pay rate is used in...
-
Austin Limited is trying to determine the value of its ending inventory as of February 28, 2014, the companys year-end. The following transactions occurred, and the accountant asked your help in...
-
Calculate the future value of the following annuities, assuming each annuity payment is made at the end of each compounding period. (FV of $1, PV of $1, FVA of $1, and PVA of $1) (Use tables, Excel,...
-
What is the value of CP's revised offer on December 8 (before CP "sweetened" its offer by adding the CVR security)? In your analysis, assume the following: a) A valuation date of December 31, 2015,...
-
Implement the following accelerated version of Euclid's algorithm for gcd(s, t) with precondition 1 < s = t, retaining s 2 , s 3 , ... s k . Diminish t by s k . If (the new) t is still the larger of...
-
pest analysis and a marketing mix for the company tui and From your market and company analysis make relevant recommendations of how the company could improve their marketing mix (price, product,...
-
Harley-Davidson is my industry/brand of interest. As you continue working on your final marketing plan in this course, pricing will be a crucial aspect of your plan. You will want to consider...
-
During its first year of operations, Eastern Data Links Corporation entered into the following transactions relating to shareholders' equity. The articles of incorporation authorized the issue of 9...
-
The ABX News Co. paid the last dividend (D 0 ) of $2/share, and the dividend is expected to grow at a rate of 12% over the next 4 years. It will then grow at a normal, constant rate of 3% for the...
-
A loan has a stated annual rate of 16.87%. If loan payments are made monthly and interest is compounded monthly, what is the effective annual rate of interest? Be sure to answer as a percentage.
-
You are going to lend a friend $5,000. You want him to repay you $7,500 in 6 years. The interest portion of his repayment is A/ Round to the nearest dollar. The simple interest rate (in percent form)...
-
The pendulum consists of two rods: AB is pin supported at A and swings only in the y-z plane, whereas a bearing at B allows the attached rod BD to spin about rod AB. At a given instant, the rods have...
-
The following information was taken from the 2009 annual report of Emerson, a leader in process management, technology, network power, and industrial automation (dollars in millions). REQUIRED...
-
Several independent transactions are as follows. 1. 10,000 Shares of no-par common stock are issued for $50 per share. 2. 10,000 Shares of $1 par value common stock are issued for $40 per share. 3....
-
Excerpts from the operating sections of the 2008 statement of cash flows for Target and Toyota are provided below. Target publishes U.S. GAAP -based financial statements and Toyota publishes...
-
Applying the criterion for equilibrium, derive the Clausius-Clapeyron equation.
-
For the system methanol (1)-methyl acetate (2), the activity coefficients for components 1 and 2 are represented by where \[ \begin{aligned} \ln \gamma_{1} & =A x_{2}^{2} \quad \text { and } \quad...
-
The pure component vapour pressure of two organic liquids \(\mathrm{X}\) and \(\mathrm{Y}\) by Antoine equations are given by \[ \ln P_{1}^{\text {Sat }}=14.35-\frac{2942}{T+220} \] and \[...

Study smarter with the SolutionInn App


