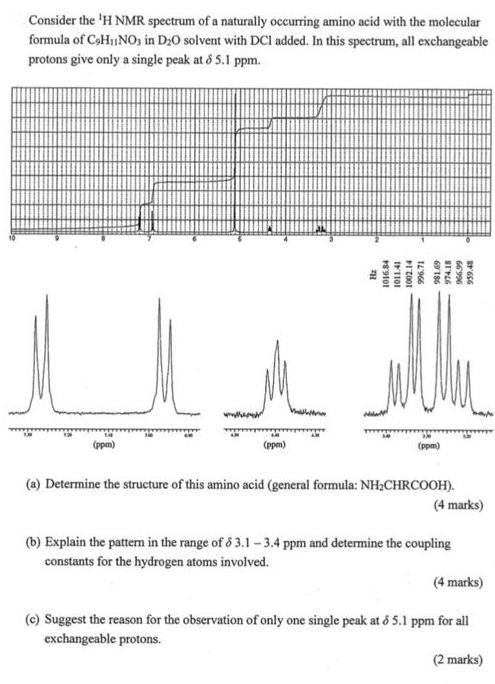
Consider the 'H NMR spectrum of a naturally occurring amino acid with the molecular formula of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the 'H NMR spectrum of a naturally occurring amino acid with the molecular formula of CoHINO, in D:0 solvent with DCl added. In this spectrum, all exchangeable protons give only a single peak at o 5.1 ppm. (ppm) (ppm) (ppm) (a) Determine the structure of this amino acid (general formula: NH;CHRCOOH). (4 marks) (b) Explain the pattem in the range of 6 3.1- 3.4 ppm and determine the coupling constants for the hydrogen atoms involved. (4 marks) (e) Suggest the reason for the observation of only one single peak at & 5.1 ppm for all exchangeable protons. (2 marks) 66996 69 186 IL966 Consider the 'H NMR spectrum of a naturally occurring amino acid with the molecular formula of CoHINO, in D:0 solvent with DCl added. In this spectrum, all exchangeable protons give only a single peak at o 5.1 ppm. (ppm) (ppm) (ppm) (a) Determine the structure of this amino acid (general formula: NH;CHRCOOH). (4 marks) (b) Explain the pattem in the range of 6 3.1- 3.4 ppm and determine the coupling constants for the hydrogen atoms involved. (4 marks) (e) Suggest the reason for the observation of only one single peak at & 5.1 ppm for all exchangeable protons. (2 marks) 66996 69 186 IL966
Expert Answer:
Answer rating: 100% (QA)
Poge Stoucture o amiluo anid 33 PpM 73PPM 69 PPM 44ppM CH Tuios... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Draw the structure of a naturally occurring amino acid that: a. contains a 1 alcohol b. contains an amide c. is an essential amino acid with an aromatic ring d. is a neutral amino acid with a 3...
-
The 1H NMR spectrum of a compound (C10H13BrO) is shown in Figure 16.10. The compound gives benzyl bromide, along with a second compound C3H6Br2, when heated with HBr. What is the first compound?
-
The 1H NMR spectrum of a compound (C10H13BrO) is shown in Figure 16.10. The compound gives benzyl bromide, along with a second compound C3H6Br2, when heated with HBr. What is the first compound?
-
The stockholders' equity accounts of Marya Corporation on January 1, 2014, were as follows. Preferred Stock (9%, $100 par, cumulative, 5,000 shares authorized) ...$300,000 Common Stock ($3 stated...
-
In Example 24.1 the data show correlation between the income and age of the customer. This produces collinearity and makes the analysis tricky to interpret. The marketing research group could have...
-
1. Was HP wise in attempting to make many major changes simultaneously? 2. What problems does it appear developed under the year 2000 reorganization? 3. What is your evaluation of Mr. Hurds approach...
-
Given the formulation of the free boundary problem for the valuation of an American Put option, \[\begin{aligned}& \frac{\partial P}{\partial t}+\frac{1}{2} \sigma^{2} S^{2} \frac{\partial^{2}...
-
Glenn Grimes is the founder and president of Heartland Construction, a real estate development venture. The business transactions during February while the company was being organized are listed...
-
Harwood Company's job-order costing system applies overhead cost to jobs based on its machine-hours. The predetermined overhead rate of $2.50 per machine-hour was based on a cost formula that...
-
Garnett Jackson, the founder and CEO of Tech Tune-Ups, stared out the window as he finished his customary peanut butter and jelly sandwich, contemplating the dilemma currently facing his firm. Tech...
-
Find a + b, 2a + 3b, lal, and la - bl. a = (3,-4), b = (-5, 2) a + b = 2a + 3b = |a| = la - bl =
-
Charged particles 1,2 , and \(3\left(q_{1}=10.0 \mu \mathrm{C}, q_{2}=-5.00 \mu \mathrm{C} ight.\), and \(\left.q_{3}=-3.00 \mu \mathrm{C} ight)\) are arranged in an equilateral triangle in an \(x...
-
You are evaluating a new product. In year 3 of your analysis, you are projecting pro forma sales of \($5\) million and cost of goods sold of \($3\) million. You will be depreciating a \($1\) million...
-
In a cylindrical chamber fitted with a piston, the piston compresses \(10.0 \mathrm{~mol}\) of ideal gas from \(1.00 \mathrm{~m}^{3}\) to \(0.100 \mathrm{~m}^{3}\) at a constant temperature of \(300...
-
Redo question 29 using a bar chart. Which method is better for presenting these election results? Question 29 The results of the 1991 city council election (voters could vote for more than one...
-
Figure Q28.5 shows the typical photoelectric behavior of a metal as the anode-cathode potential difference \(\Delta V\) is varied. a. Why do the curves become horizontal for \(\Delta V \geqq 1...
-
Spencer would like to purchase a new car for $12,000. How much will his monthly payments be if he took out a 3-year loan at 7% interest? Show your work.
-
Grace is training to be an airplane pilot and must complete five days of flying training in October with at least one day of rest between trainings. How many ways can Grace schedule her flying...
-
The compound having the structure shown was heated with dilute sulfuric acid to give a product having the molecular formula C5H12O3 in 63-71% yield. Propose a reasonable structure for this product....
-
Compounds known as lactones, which are cyclic esters, are formed on Baeyer-Villiger oxidation of cyclic ketones. Suggest a mechanism for the Baeyer-Villiger oxidation shown. C.H.CO OH Cyclopentanone...
-
Verify that the formal charges on nitrogen in ammonium ion and boron in borohydride ion are as shown.
-
Assume the same facts as in Problem I:9-53, except that Becky and Ken are not related and that under the terms of the loan Ken agrees to repay Becky the \($5,000\) plus interest (at a reasonable...
-
During 2022, Becky loans her brother Ken \($5,000,\) which he intends to use to establish a small business. Because Ken has no other assets and needs cash to establish the business, the agreement...
-
During 2023, Pam incurred the following casualty losses: All of the items were destroyed in the same casualty, which resulted from a federally declared disaster. Before considering the casualty...

Study smarter with the SolutionInn App


