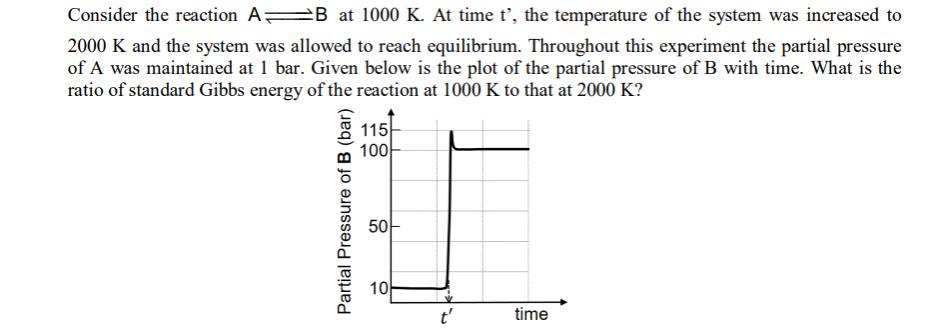
Consider the reaction AB at 1000 K. At time t', the temperature of the system was...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the reaction AB at 1000 K. At time t', the temperature of the system was increased to 2000 K and the system was allowed to reach equilibrium. Throughout this experiment the partial pressure of A was maintained at 1 bar. Given below is the plot of the partial pressure of B with time. What is the ratio of standard Gibbs energy of the reaction at 1000 K to that at 2000 K? Partial Pressure of B (bar) 115 100 50 10 t' time Consider the reaction AB at 1000 K. At time t', the temperature of the system was increased to 2000 K and the system was allowed to reach equilibrium. Throughout this experiment the partial pressure of A was maintained at 1 bar. Given below is the plot of the partial pressure of B with time. What is the ratio of standard Gibbs energy of the reaction at 1000 K to that at 2000 K? Partial Pressure of B (bar) 115 100 50 10 t' time
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Consider the following vapor pressure versus temperature plot for three different substances A, B, and C. If the three substances are CH4, SiH4, and NH3, match each curve to the correct substance. A...
-
At 2000 oC the equilibrium constant for the reaction Is Kc = 2.4 Ã 103. If the initial concentration of NO is 0.175 M, what are the equilibrium concentrations of NO, N2, and O2?
-
The equilibrium reaction with methane as CH4 C + 2H2 has ln K = -0.3362 at 1440 R and ln K = -4.607 at 1080 R. By noting the relation of K to temperature, show how you would...
-
If the rate of a plane in still air is x mph and the rate of a steady wind is 20 mph, what is the rate of the plane in each case? (a) The plane is flying into the wind (that is, into a headwind,...
-
Round Tire Co. sells 1,000 tires to Green Rent-a-Car for use on Greens fleet. The same day, it sells one new tire to Betty Blue for use on her car. For both sales, Round uses a sales agreement that...
-
A review of the ledger of Gina Company at December 31, 2020, produces the following data pertaining to the preparation of annual adjusting entries. 1. Prepaid Insurance $10,440. The company has...
-
Explain how organizations should choose appropriate T&D methods.
-
Yancey Co. receives $300,000 when it issues a $300,000, 10%, mortgage note payable to finance the construction of a building at December 31, 2017. The terms provide for annual installment payments of...
-
Prepare the four required closing entries for a company that has the following account balances. Capital Stock 500,000 Rent Revenue 5/s 200,000 Fees Earned 100,000 325,000 300,000 Wage Expense...
-
Jay Gatsby categorizes wines into one of three clusters. The centroids of these clusters, describing the average characteristics of a wine in each cluster, are listed in the following table. Jay has...
-
Obtain the transfer functions X(s)/F(s) and Y(s)/F(s) for the following model: 3x=8y y=2f(t)-2y-3x
-
Find the points where the two curves intersect. List your answers as points in the form (x, y). If there are no solutions, type "None". Answer (separate by commas): (f(x) = x-3x+ g(x) = x+1 3x+4)
-
According to the principle of separation of Betty Brown's ballots, there will be a total number of ballots to be formed along with 1 ballot to hold a seat in the Board of Directors. Therefore, there...
-
Tobesco Corporation 2020 and 2021 Balance sheets (in millions) 2020 2021 Change Assets Current assets Cash 84 146 62 Accounts receivable 165 188 23 Inventory 393 422 29 Total current assets 642 756...
-
Halo Enterprises recently experienced a fire, forcing the company to use incomplete information to analyze operations. Consider the following data and assume that all materials purchased during the...
-
In the dynamic landscape of manufacturing and sales, Caribann is a company with the potential to produce 100,000 units of its sole product annually. Caribann's interplay of costs and production...
-
Assume that the market for gasoline can be represented by the following demand and supply functions: QD = -2P+24, and Qs = 10P, where P is the price per gallon, and QD and Qs each represents quantity...
-
A routine activity such as pumping gasoline can be related to many of the concepts studied in this text. Suppose that premium unleaded costs $3.75 per gal. Work Exercises in order. Use the...
-
Why are d orbitals sometimes used to form hybrid orbitals? Which period of elements does not use d orbitals for hybridization? If necessary, which d orbitals (3d, 4d, 5d, or 6d) would sulfur use to...
-
From the values in Table for the van der Waals constant a for the gases H 2 , CO 2 , N 2 , and CH 4 , predict which molecule shows the strongest intermolecular attractions.Table \ 7 1 200 1 6 1 8 2...
-
The binding energy per nucleon for magnesium-27 is 1.326 X 10-12 J/nucleon. Calculate the atomic mass of magnesium-27.
-
Bethany, who weighs 560 N, lies in a hammock suspended by ropes tied to two trees. One rope makes an angle of 45 with the ground; the other makes an angle of 30. Find the tension in each of the ropes.
-
In the Skycoaster amusement park ride, riders are suspended from a tower by a long cable. A second cable then lifts them until they reach the starting position indicated in Figure P5.3. The lifting...
-
A dump truck, whose bed is made of steel, holds an old steel watering trough. The bed of the truck is slowly raised until the trough begins to slide. What is the acceleration of the trough as it...

Study smarter with the SolutionInn App



