Consider the titration described here: 0.7917 g of anhydrous sodium thiosulphate (Na 2 S 2 O 3
Fantastic news! We've Found the answer you've been seeking!
Question:
Consider the titration described here:
- 0.7917 g of anhydrous sodium thiosulphate (Na 2 S 2 O 3 ), previously purified and dried in an oven, is weighed into an erlenmeyer flask.
- This sample is titrated with a mixture of KIO 3 and KI that contains approximately 0.1 M I 3 - . This titration requires 23.56 mL of titrant to reach the endpoint (found with a starch indicator).
- This same mixture of KIO 3 and Kl is then used to titrate a solution containing an unknown amount of ascorbic acid. This titration requires 37.21 mL of titrant to reach the endpoint (found with a starch indicator).
- The ascorbic acid sample used in the titration above was prepared by using a 25 mL pipet to deliver an aliquot of the solution into an erlenmeyer flask.
Relevant reactions:
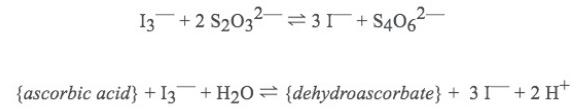
Based on this information, what is the concentration (in mol/L) of ascorbic acid in the "unknown" liquid?
Related Book For 

Posted Date:




