Color and absorption spectra. Color Plate 15 shows colored solutions and their spectra. From Table 17-1, predict
Question:
Color and absorption spectra. Color Plate 15 shows colored solutions and their spectra. From Table 17-1, predict the color of each solution from the wavelength of maximum absorption. Do observed colors agree with predicted colors?
Table 17-1
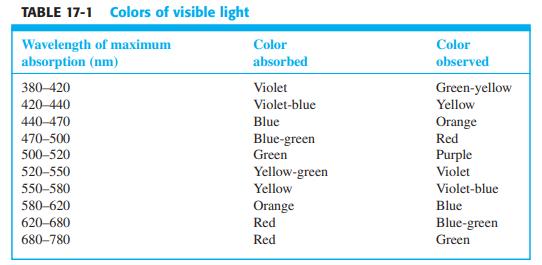
TABLE 17-1 Colors of visible light Wavelength of maximum absorption (nm) Color Color absorbed observed 380-420 Violet Green-yellow 420-440 Violet-blue Yellow 440-470 Blue Orange 470-500 500-520 Red Blue-green Green Purple 520-550 Yellow-green Violet 550-580 Yellow Violet-blue 580-620 Orange Blue 620-680 Red Blue-green 680-780 Red Green
Step by Step Answer:
Curve A B D E F Absorption peak nm 760 ...View the full answer



Related Video
Using a few basic physics principles, you can impress your friends with a trick that makes a bottle disappear. For this experiment, you will need a mini plastic bottle, glycerin, a glass, and a funnel. First, open the bottle and pour some glycerin into it, then close the bottle tightly. Next, pour some water into the glass using a funnel. Place the mini bottle into the glass of water and it will look normal. This is because light travels through air faster than it travels through the glass and water, allowing our eyes to see the bottle inside the glass. However, when you fill the mini bottle with more glycerin, and pour glycerin into the glass, then put the glycerin filled bottle into the glass with glycerin in it. Half of the bottle that is submerged in the glycerin will become invisible as the light travels through glass and glycerin at the same speed, thus it does not bend and no refraction takes place, making the bottle invisible. This happens because both glass and glycerin have almost the same refractive index, which causes the speed of light to be the same in both mediums, causing no bending of light and making the bottle disappear.
Students also viewed these Chemical Engineering questions
-
The photograph of upconversion in Color Plate 19 shows total internal reflection of the blue ray inside the cuvet. The angle of incidence of the blue ray on the wall of the cuvet is 55. We estimate...
-
The solutions shown here each have an absorption spectrum with a single absorption peak like that shown in Figure 23.26. What color does each solution absorb most strongly? [Section 23.5]
-
Predict the positions of the absorption bands in the IR spectra for the carbonyl groups of thesecompounds. HO. b) CH,CH-CHCH (p
-
One major concern about the future is that water scarcity will grow, particularly in arid regions where precipitation levels may be reduced by climate change. Will our institutions provide for an...
-
In a recent newspaper release, the president of Downs Company asserted that something has to be done about depreciation. The president said, Depreciation does not come close to accumulating the cash...
-
The GNG Café, a new concept in on-campus dining features homemade bakery items, upscale sandwiches and wraps, fresh salads, and signature soups. The modern café-style design...
-
Explain synchronous and asynchronous communication in relation to elearning.
-
Creative Solutions purchased a patent from Russell Lazarus, an inventor. At the time of the purchase, the patent had two years remaining. The president of Creative Solutions decided to have the...
-
Presented below is information related to Sunbuck's Coffee Club for its fiscal year ending April 30, 2023. During the year, the Board of Directors decided to discontinue the operations of its entire...
-
On January 1, 2019, Concord Corp. signs a contract to lease nonspecialized manufacturing equipment from Stone Inc. Concord agrees to make lease payments of $47,500 per year. Additional information...
-
Characteristic orange light produced by sodium in a flame is due to an intense emission called the sodium D line, which is actually a doublet, with wavelengths (measured in vacuum) of 589.157 88 and...
-
This problem can be worked by calculator or with the spreadsheet in Figure 18-5. Consider compounds X and Y in the example labeled "Analysis of a Mixture, Using Equations 18-6" in Section 18-1. Find...
-
Define producer surplus. Draw a graph to show the area that represents producer surplus.
-
Interview at least ten people and ask the following questions: What are your auto liability insurance limits and deductibles? If you own your own home, do you have an all-risk or named perils...
-
Lee Chiew is borrowing \($170\),000 from Asia Bank USA to open Asian Health Foods in Virginia. She agrees to repay the loan at 10 percent compounded annually. Since she expects it to take a few years...
-
Solve the IVP. Do not use Laplace transformation. \(\dot{y}=(1-y) \sin t, y\left(\frac{1}{2} \pi ight)=2\)
-
Do a Google search on something like home loans; mortgage lenders; home financing; fixed loans; ARM and spend 10 minutes seeing what you can learn about available home loans. Give the URL of any Web...
-
A series of 10 end-of-year deposits is made that begins with $7,000 at the end of year 1 and decreases at the rate of $300 per year with 10 percent interest. a. What amount could be withdrawn at t =...
-
Relative to the stars, the moon takes 27*1/3 days to orbit the earth. As seen from the earth, the moon drifts eastward relative to the stars. Through what angle does the moon move eastward each day...
-
Briefly discuss the implications of the financial statement presentation project for the reporting of stockholders equity.
-
Consider the following reaction: 2 NO(g) + 2 H2(g) -- N2(g) + 2 H2O(g) (a) The rate law for this reaction is first order in H2 and second order in NO. Write the rate law. (b) If the rate constant for...
-
Consider the following reaction: CH3Br(aq) + OH (aq) -- CH3OH(aq) + Br(aq) The rate law for this reaction is first order in CH3Br and first order in OH. When [CH3Br] is 5.0 10-3 M and [OH] 0.050 M,...
-
The reaction between ethyl bromide (C2H5Br) and hydroxide ion in ethyl alcohol at 330 K, C2H5Br(alc) + OH (alc) -- C2H5OH(I) + Br(aIc), is first order each in ethyl bromide and hydroxide ion. When...
-
Have a brief introduction, discussion of the topics/points, and a conclusion.what you have learned during the course( Organizational Dynamics and Effectiveness).Their relevance to your career....
-
Using the article provided below, you are required to analyse the company information to define the business goals and evaluate whether the supply chain is a strategic fit with the business and...
-
Create output that shows a scatterplot with a line of best fit for your data. Explain what a line of best fit is? Submit both the output file (SPSS) as well as your Word summary.

Study smarter with the SolutionInn App


