contacts the top of the cylinder A, the cylinder Just after the brake arm B in...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
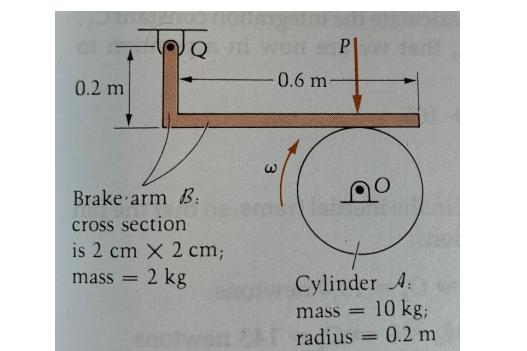
contacts the top of the cylinder A, the cylinder Just after the brake arm B in the figure is turning clockwise at 1000 rpm. The coefficient of kinetic friction between A and B is u = 0.3. Find how long it takes for A to come to rest under the constant force P = 40 kN. Consider the brake arm B to be in equilibrium (does not move). The total mass of the brake arm is 2 kg: 0.5 kg for the vertical part, and 1.5 kg for the horizontal part. The horizontal distance between Q and P is 0.401m (due to the width of the arm). Assume pins Q and O to be frictionless. For the cylinder A: Io = ½ mr², with m mass and r radius. 0.2 m TOUST Brake arm B: cross section is 2 cm x 2 cm; 2 kg mass = 0.6 m- PI Cylinder A: mass= 10 kg; radius= 0.2 m contacts the top of the cylinder A, the cylinder Just after the brake arm B in the figure is turning clockwise at 1000 rpm. The coefficient of kinetic friction between A and B is u = 0.3. Find how long it takes for A to come to rest under the constant force P = 40 kN. Consider the brake arm B to be in equilibrium (does not move). The total mass of the brake arm is 2 kg: 0.5 kg for the vertical part, and 1.5 kg for the horizontal part. The horizontal distance between Q and P is 0.401m (due to the width of the arm). Assume pins Q and O to be frictionless. For the cylinder A: Io = ½ mr², with m mass and r radius. 0.2 m TOUST Brake arm B: cross section is 2 cm x 2 cm; 2 kg mass = 0.6 m- PI Cylinder A: mass= 10 kg; radius= 0.2 m
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Does the number of years that a woman is on birth control (X) have an effect on how long it takes for her to become pregnant (Y) once she ceases to use it and tries to have children? A curious...
-
At each point evaluate the function or indicate that the function is undefined there. k(x,y) = xe, (1,0), (3,3), (0, 12)
-
As the level of output increases, what happens to the difference between the value of average total cost and the value of average variable cost?
-
Are nursing salaries in Tampa, Florida, lower than those in Dallas, Texas? Salary data show staff nurses in Tampa earn less than staff nurses in Dallas (The Tampa Tribune, January 15, 2007). Suppose...
-
Blockbuster, Inc., is the world's leading provider of rentable DVDs and videogames. Blockbuster estimates that 64 percent of the U.S. population lives within a 10-minute drive of a Blockbuster store....
-
A piece of equipment that cost $32,400 and on which $18,000 of accumulated depreciation had been recorded was disposed of on January 2, the first day of business of the current year. For each of the...
-
Why is Microsoft Corp bond a good investment? The Bond has a maturity date of 6/1/2039 and a coupon rate of 5.2000% and is At the current price of 114.234 USD, this equates to a 4.16 percent annual...
-
Find horizontal and Vertical components of zipline, Enter your answer in terms of T, theta_ R for Right theta angle; theta_ L for Left theta angle, v_x, v_y, c, m and g. Modeling the rider as a point...
-
Loss Company had losses of $100,000 to carry forward. In the loss year it had the following shareholders with the following shareholding: A 25% B 25% C 25% D 25% In the gain year, it had assessable...
-
Discuss the stability of different conformations of n-butane (use Newman projections) with the help of their energy level diagram.
-
A 5.0 liter sample of argon gas is collected at 40C and 700 torr pressure. Select the proper set-up for the calculation of the STP volume that the gas will occupy. PV/T= P2V2/T2 and V = PVT2/PT1 STP...
-
If 3.50x1014 kg of CO are cycled through the biosphere annually, how many people could be produced each year from this amount of CO? (Assume that the average person is 70.0 kg and composed of 18 %...
-
If 16.5 g of NaOH is added to 0.750 L of 1.00 M Cd (NO3)2, how many grams of Cd (OH)2 will be formed in the following precipitation reaction? 2 NaOH(aq) + Cd (NO3)2(aq) Cd (OH)2 (s) + 2 NaNO3(aq)
-
An electron in a periodic lattice has the following energy-wavenumber relationship: E (k) 2m cos (k) a. Plot the E-k diagram (0.5p) b. Determine the effective mass (0.5p) c. Determine the group...
-
Which of the following statements is incorrect in relation to disclosure of balances for property, plant and equipment? a. A summary of movements in the revaluation surplus is required to be...
-
H Corporation has a bond outstanding. It has a coupon rate of 8 percent and a $1000 par value. The bond has 6 years left to maturity but could be called after three years for $1000 plus a call...
-
Calculate the mass of the Sun based on data for Earths orbit and compare the value obtained with the Suns actual mass.
-
Find the ratio of the diameter of aluminum to copper wire, if they have the same resistance per unit length (as they might in household wiring).
-
Calculate the voltage applied to a 2.00 F capacitor when it holds 3.10 C of charge.
-
Imagine that a company is converting from conventional mass technology to a highly flexible, computerized, integrated production system. List structural and behavioural problems that the company...
-
Discuss this statement: The effects of advanced information technology on job design and organizational structure are highly predictable.
-
Distinguish among pooled interdependence, sequential interdependence, and _ reciprocal interdependence in terms of the key problem each poses for organizational effectiveness.

Study smarter with the SolutionInn App


