Crane Industries has three activity cost pools and two products. It expects to produce 3,200 units...
Fantastic news! We've Found the answer you've been seeking!
Question:
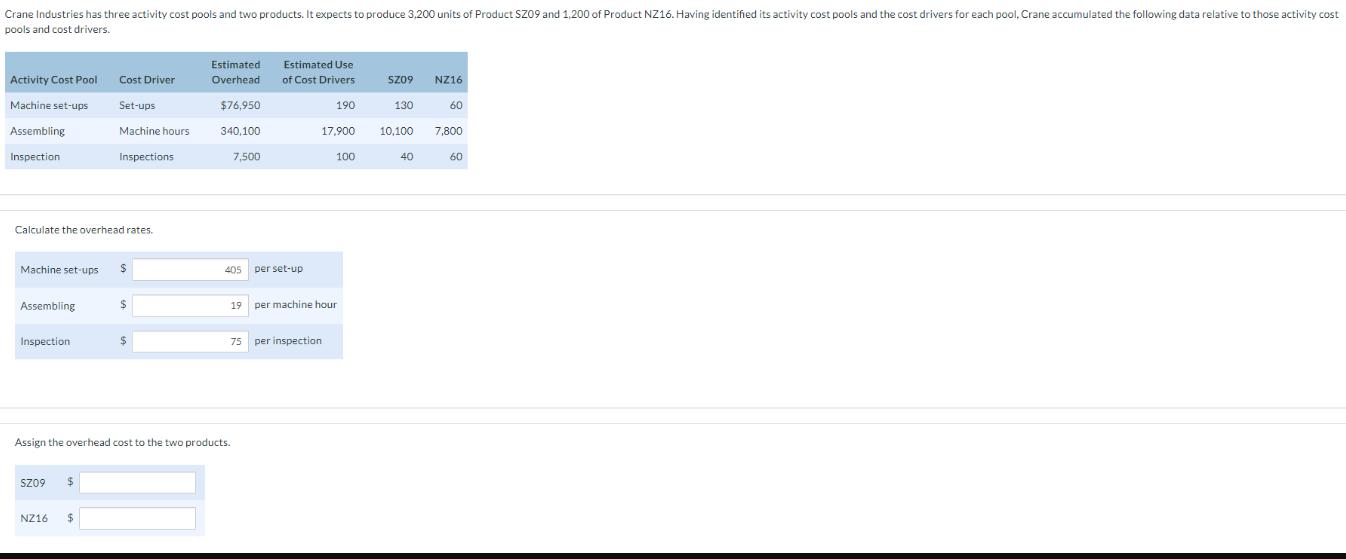
Transcribed Image Text:
Crane Industries has three activity cost pools and two products. It expects to produce 3,200 units of Product SZ09 and 1,200 of Product NZ16. Having identified its activity cost pools and the cost drivers for each pool, Crane accumulated the following data relative to those activity cost pools and cost drivers. Activity Cost Pool Machine set-ups Assembling Inspection Machine set-ups Calculate the overhead rates. Assembling Inspection Cost Driver Set-ups Machine hours SZ09 $ Inspections NZ16 $ $ $ $ $ Estimated Overhead $76,950 340,100 7,500 405 Assign the overhead cost to the two products. Estimated Use of Cost Drivers per set-up 75 per inspection 190 17,900 19 per machine hour 100 SZ09 130 NZ16 40 60 10,100 7,800 60 Crane Industries has three activity cost pools and two products. It expects to produce 3,200 units of Product SZ09 and 1,200 of Product NZ16. Having identified its activity cost pools and the cost drivers for each pool, Crane accumulated the following data relative to those activity cost pools and cost drivers. Activity Cost Pool Machine set-ups Assembling Inspection Machine set-ups Calculate the overhead rates. Assembling Inspection Cost Driver Set-ups Machine hours SZ09 $ Inspections NZ16 $ $ $ $ $ Estimated Overhead $76,950 340,100 7,500 405 Assign the overhead cost to the two products. Estimated Use of Cost Drivers per set-up 75 per inspection 190 17,900 19 per machine hour 100 SZ09 130 NZ16 40 60 10,100 7,800 60
Expert Answer:
Related Book For 

Accounting Tools for business decision making
ISBN: 978-0470095461
4th Edition
Authors: kimmel, weygandt, kieso
Posted Date:
Students also viewed these accounting questions
-
Flynn Industries has three activity cost pools and two products. It expects to produce 3,000 units of Product BC113 and 1,500 of Product AD908. Having identified its activity cost pools and the cost...
-
Weber Industries has three activity cost pools and two products. It expects to produce 3,000 units of Product BC113 and 1,400 of Product AD908. Having identified its activity cost pools and the cost...
-
Weber Industries has three activity cost pools and two products. It expects to produce 3,000 units of Product BC113 and 1,400 of Product AD908. Having identified its activity cost pools and the cost...
-
South Sea Baubles has the following (incomplete) balance sheet and income statement. a. What is shareholders' equity in 2013 and 2014? b. What is net working capital in 2013 and 2014? c. What are...
-
Financial performance is measured in many ways. Requirements 1. Explain the difference between lag and lead indicators. 2. The following is a list of financial measures. Indicate whether each is a...
-
Name the following compounds: a. HCl(g) b. HBr(aq) c. HF(g) d. HNO 3 (aq)
-
What does chance have to do with the testing of the research hypothesis for significance?
-
The stockholders equity accounts of Ashley Corporation on January 1, 2012, were as follows. Preferred Stock (8%, $50 par, cumulative, 10,000 shares authorized)..... $ 400,000 Common Stock ($1 stated...
-
What advice would you give an incoming CEO? In 2005, what did Advent International and Highland Capital see in lululemon? How important is lululemon's culture? Why was Bob Meers hired I 2005? What...
-
The G. Saussy Manufacturing company is putting out four new electronic components. Each of Saussy's four plants has the capacity to add one more product to its current line of electronic parts. The...
-
For the UNM homecoming game, there a total of 26,182 tickets sold. The price for students were $7 and the price for adults were $12. The total sales were $220,379. How many student tickets were sold...
-
How to create on Android Studio using JAVA? About this assignment In this assignment, I would like you to create an app to introduce a bit about yourself (see below as an example). You can write...
-
Develop a C# console application that simulates a vending machine. Write a method in C# that converts a number from binary to decimal format. Create a simple To-Do List desktop application using...
-
Crystal Limited manufactures photo frames. The photo frames are assembled in the Frame Division. The half-finished frames are then transferred to the Finishing Division, where the glass and other...
-
Oregon Corporation has filed a voluntary petition to reorganize under Chapter 11 of the Bankruptcy Reform Act. Its creditors are considering an attempt to force liquidation. The company currently...
-
Overview: This journal activity is private between you and the instructor. This assignment will allow you to assess the impact of news on stocks and bonds. News can affect stock and bond prices in...
-
Use the information below to answer the questions that follow. The business's year-end is December 31. Cost of equipment = 60000 Useful life in years = 5 Residual value = 0 Date purchased = August 1,...
-
Stephen Schor, an accountant in New York City, advised his client, Andre Romanelli, Inc., to open an account at J. P. Morgan Chase Bank, N.A., to obtain a favorable interest rate on a line of credit....
-
Using the data in BE22-6 and BE22-10, compute the overhead volume variance. Normal capacity was 25,000 direct labor hours. Data from BE22-10 In October, Derby Company reports 21,000 actual direct...
-
American Products Corporation participates in a highly competitive industry. In order to meet this competition and achieve profit goals, the company has chosen the decentralized form of organization....
-
Gwyn Wallander is the new owner of Bennett Co. She has heard about internal control but is not clear about its importance for her business. Explain to Gwyn the four purposes of internal control, and...
-
For liquid acetone at \(20^{\circ} \mathrm{C}\) and \(1 \mathrm{bar}\), \[ \beta=1.487 \times 10^{-3} /{ }^{\circ} \mathrm{C} \quad \alpha=62 \times 10^{-6} / \mathrm{bar} \quad V=1.287...
-
Determine the molar volume of ammonia vapour and ammonia liquid at \(321.55 \mathrm{~K}\) and 1.95 \(\mathrm{MPa}\). Ammonia is assumed to follow van der Waals equation of state.
-
Any equation that relates to the pressure, temperature and volume is called an equation of state. Justify the statement.

Study smarter with the SolutionInn App



