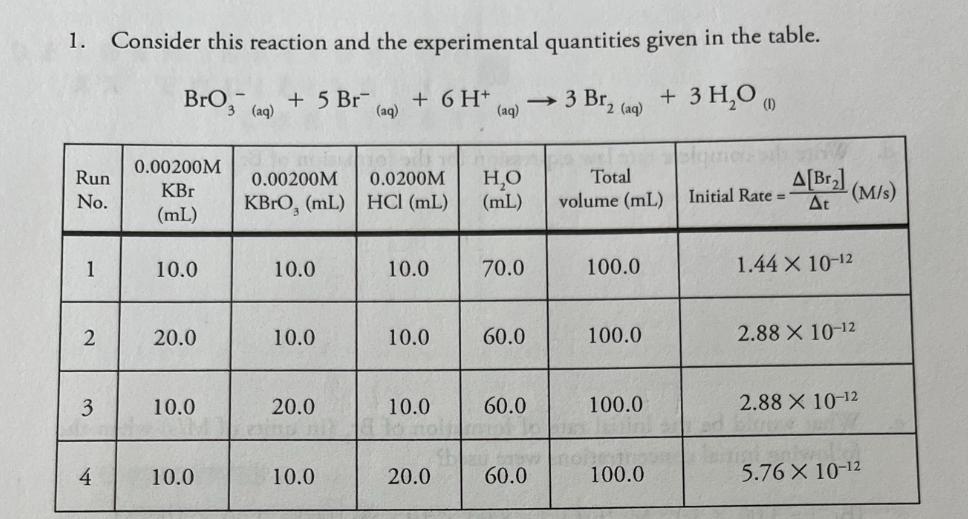
+ 5 Br (aq) 0.00200M 1. Consider this reaction and the experimental quantities given in the...
Fantastic news! We've Found the answer you've been seeking!
Question:


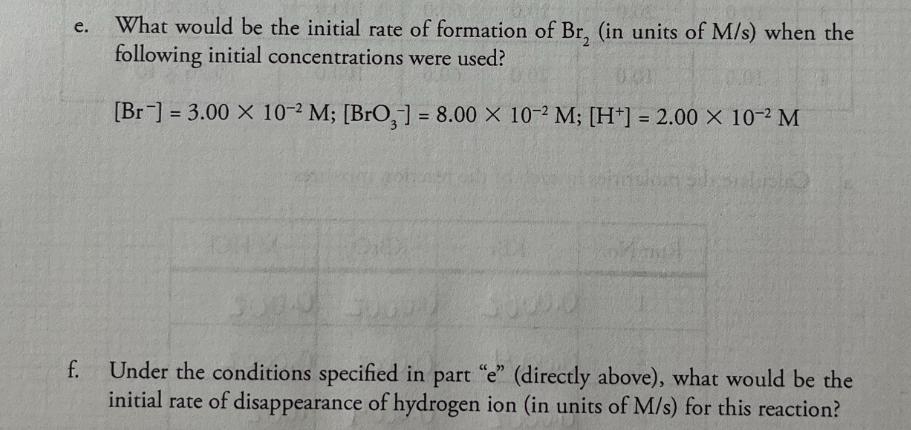
Transcribed Image Text:
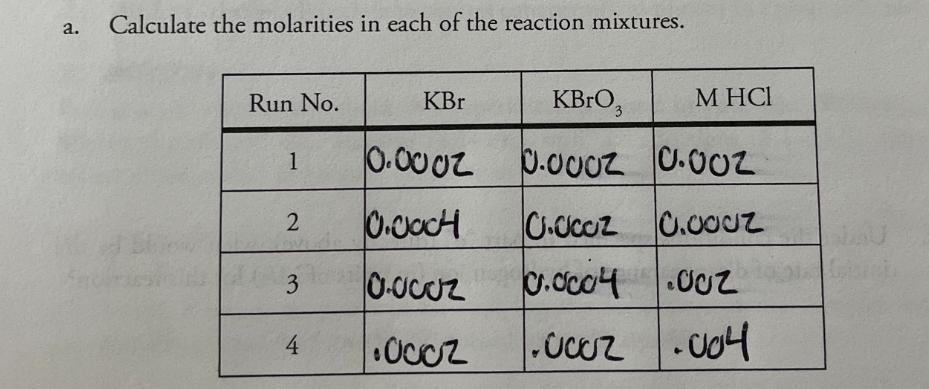
+ 5 Br (aq) 0.00200M 1. Consider this reaction and the experimental quantities given in the table. BrO3 (aq) + 6 H+ (aq) 3 Br, 2 (aq) + 3 H₂O (1) Run 0.00200M 0.0200M H₂O Total KBr No. KBrO, (mL) HCl (mL) (mL) volume (mL) Initial Rate= A[Br₂] At (M/s) (mL) 1 10.0 10.0 10.0 70.0 100.0 1.44 X 10-12 2 20.0 10.0 10.0 60.0 100.0 2.88 X 10-12 3 10.0 20.0 10.0 60.0 100.0 2.88 X 10-12 to hol 4 10.0 10.0 20.0 60.0 100.0 5.76 X 10-12 a. Calculate the molarities in each of the reaction mixtures. Run No. KBr KBrO3 M HCl 1 0.0002 0.0002 0.002 2 0.0004 0.0002 0.0002 3 0.0002 0.0004 .002 0.0004.002 4 .0002 .0002 .004 b. Determine the order of each reactant. (Show work below.) order of [Br] order of [BrO3] order of [H+] C. Determine the value of the rate constant (including units) for the formation of Br₂. d. Write the complete rate law expression for the formation of Br.. e. What would be the initial rate of formation of Br, (in units of M/s) when the following initial concentrations were used? [Br] =3.00 x 10-2 M; [BrO,] = 8.00 x 10-2 M; [H+] = 2.00 x 10-2 M f. Under the conditions specified in part "e" (directly above), what would be the initial rate of disappearance of hydrogen ion (in units of M/s) for this reaction? + 5 Br (aq) 0.00200M 1. Consider this reaction and the experimental quantities given in the table. BrO3 (aq) + 6 H+ (aq) 3 Br, 2 (aq) + 3 H₂O (1) Run 0.00200M 0.0200M H₂O Total KBr No. KBrO, (mL) HCl (mL) (mL) volume (mL) Initial Rate= A[Br₂] At (M/s) (mL) 1 10.0 10.0 10.0 70.0 100.0 1.44 X 10-12 2 20.0 10.0 10.0 60.0 100.0 2.88 X 10-12 3 10.0 20.0 10.0 60.0 100.0 2.88 X 10-12 to hol 4 10.0 10.0 20.0 60.0 100.0 5.76 X 10-12 a. Calculate the molarities in each of the reaction mixtures. Run No. KBr KBrO3 M HCl 1 0.0002 0.0002 0.002 2 0.0004 0.0002 0.0002 3 0.0002 0.0004 .002 0.0004.002 4 .0002 .0002 .004 b. Determine the order of each reactant. (Show work below.) order of [Br] order of [BrO3] order of [H+] C. Determine the value of the rate constant (including units) for the formation of Br₂. d. Write the complete rate law expression for the formation of Br.. e. What would be the initial rate of formation of Br, (in units of M/s) when the following initial concentrations were used? [Br] =3.00 x 10-2 M; [BrO,] = 8.00 x 10-2 M; [H+] = 2.00 x 10-2 M f. Under the conditions specified in part "e" (directly above), what would be the initial rate of disappearance of hydrogen ion (in units of M/s) for this reaction?
Expert Answer:
Answer rating: 100% (QA)
b To determine the order of each reactant we can use the method of initial rates By comparing the in... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
describe How do mineral salts participate in osmotic regulation?
-
Sewing industries is a factory located in Divundu that specialises in assembling mini-sewing electrical machines which are portable and which can be used in close proximity. The business has provided...
-
Watch the film & quot; Half the Sky & quot; and write a paper reflection of film in relation to readings, discussions and community service for this course.
-
Draw several field lines surrounding the three charged particles shown in Figure P24.10. Data from Figure P24.10 Figure P24.10 +q -29 +q
-
Elliott started business on 1 July 2025. The accounting system includes a sales journal, a purchases journal, a cash receipts journal, a cash payments journal and a general journal. The chart of...
-
Jason Trevor owns a commercial bakery in Blakely, Georgia, that produces a variety of goods sold in grocery stores. Trevor is required by law to perform internal tests on food produced at his plant...
-
Handshake owns and manages a small manufacturing business in Narok town. The following balances have been extracted from his books of account at 31 December 2018: Dr Cr Sh. Sh. Capital 1,711,200...
-
Lox, Stock and Bagel Company (LSB) is determining its cost of capital. It uses a risk free, medium term bank loan and equity financing. Part a: The current balance on the bank loan is $8,000,000. It...
-
One of Vista plc's trade investments was sold for a loss of 3.7 million, which was included in the company's income statement just below profit after tax. To arrive at profit or loss before taxes,...
-
"In years when people buy many shares of stock, investment will be high and, therefore, so will GDP." Explain in maximum of 3 lines why the statement is right or wrong. [3 marks]
-
A machine was purchased for a sum of 393,000$ having useful life of 13 years. How to account the level of depreciation under Sum of years Digits Method? Note: Salvage Value =39,000$. Brief the final...
-
On your way back home, you notice that almost every store is closed. Your little sister says, "It looks like every business is struggling because of COVID-19." You point that most events result in...
-
There are 6 rational pirates, 1, 2, 3, 4, 5 and 6. They found 242 gold coins and they must decide how to distribute them. The pirates have a strict order of seniority: pirate 1 is senior to pirate 2,...
-
1. Write a Newton-Raphson root solver 2. Write a bi-section root solver 3. Write a secant method root-solver 4. Demonstrate their effectiveness on the following functions. Your solution should...
-
What is the pH of a buffer solution that is prepared by dissolving 25.5 g of sodium acetate in a sufficient volume of 0.550 M acetic acid to obtain 500 ml of buffer, knowing that its Ka is 1.80x10-5?
-
An auto-parts manufacturer is considering establishing an engineering computing center. This center will be equipped with three engineering workstations each of which would cost $25,000 and have a...
-
Several years ago, Revnon acquired a 30% interest in Aumet at book value. During 2012 and 2013, intercompany sales of merchandise amounted to $120,000 and $180,000. On December 31, 2012, and December...
-
Logan regularly sells inventory to its 100% owned subsidiary, Newton. It sells at a gross profit of 40%. Newton sells inventory to its customers at a gross profit of 30%. Logan pays tax at a rate of...
-
It is Monday, September 16, 2013. You, CA, work at Fahmy & Gingras LLP, a CA ï¬rm. Ken Ndiaye, one of the partners, approaches you mid-morning regarding Brennan & Sons Limited (BSL), a...
-
Which is an advantage to an employee who participates in a profit-sharing plan? A. Employee does not have to make investment decisions. B. Graded vesting schedule. C. Older employees receive the...
-
Which of the following is a defined benefit plan? A. 403(b). B. 412(i). C. 457(b). D. 457(f).
-
An SEP has which characteristic? A. Loans and hardship withdrawals are available. B. Age-weighting or cross-testing is permitted. C. Social Security integration is permitted. D. Employer matching is...

Study smarter with the SolutionInn App


