Lisali Company gathered the following information related to inventory that it owned on December 31, 2015:...
Fantastic news! We've Found the answer you've been seeking!
Question:
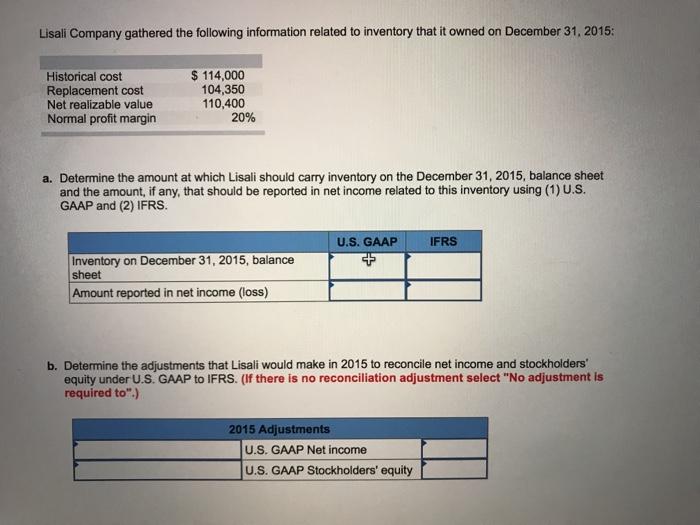
Transcribed Image Text:
Lisali Company gathered the following information related to inventory that it owned on December 31, 2015: $ 114,000 104,350 110,400 20% Historical cost Replacement cost Net realizable value Normal profit margin a. Determine the amount at which Lisali should carry inventory on the December 31, 2015, balance sheet and the amount, if any, that should be reported in net income related to this inventory using (1) U.S. GAAP and (2) IFRS. U.S. GAAP IFRS Inventory on December 31, 2015, balance sheet Amount reported in net income (loss) b. Determine the adjustments that Lisali would make in 2015 to reconcile net income and stockholders equity under U.S. GAAP to IFRS. (If there is no reconciliation adjustment select "No adjustment is required to".) 2015 Adjustments U.S. GAAP Net income U.S. GAAP Stockholders' equity Lisali Company gathered the following information related to inventory that it owned on December 31, 2015: $ 114,000 104,350 110,400 20% Historical cost Replacement cost Net realizable value Normal profit margin a. Determine the amount at which Lisali should carry inventory on the December 31, 2015, balance sheet and the amount, if any, that should be reported in net income related to this inventory using (1) U.S. GAAP and (2) IFRS. U.S. GAAP IFRS Inventory on December 31, 2015, balance sheet Amount reported in net income (loss) b. Determine the adjustments that Lisali would make in 2015 to reconcile net income and stockholders equity under U.S. GAAP to IFRS. (If there is no reconciliation adjustment select "No adjustment is required to".) 2015 Adjustments U.S. GAAP Net income U.S. GAAP Stockholders' equity
Expert Answer:

Related Book For 

Intermediate Financial Management
ISBN: 978-1285850030
12th edition
Authors: Eugene F. Brigham, Phillip R. Daves
Posted Date:
Students also viewed these accounting questions
-
The Verbrugge Publishing Company's 2015 balance sheet and income statement are as follows (in millions of dollars): Balance Sheet Income Statement Net sales...
-
The GASB requires that information be reported in certain sections of the CAFR. Following is a list of items that could appear in a CAFR report. 1. Budgetary schedules 2. Letter of transmittal 3....
-
What are the particular requirements that must be reported in a strategic report?
-
Revenue Recognition Alternatives Each of the following independent situations relates to the recognition of revenue: a. Interest on loans made by a bank b. Interest on loans made by a bank when the...
-
Give full details of the three-phase commit protocol in a distributed environment. Outline the algorithms for both coordinator and participants.
-
Assume the role of Gene Harley, the leasing manager of Northern Shopping Plaza. You have decided not to renew the lease of T-shirts Plus, which operates a tiny T-shirt decorating outlet in the mall....
-
Using the fourth-order Runge-Kutta method, solve Problem 11.15. Data From Problem 11.15:- Using the second-order Runge-Kutta method, solve the differential equation \(\ddot{x}+1000 x=0\) with the...
-
(Multiple Choice) 1. Which of the following is classified as an accrued payroll liability? Federal Income Employees Share Tax Withheld of F.I.C.A. Taxes a. No ............Yes b. No ............ No c....
-
What is the relationship between diversity, inclusion, and mental health in the workplace, and how can organizations create supportive environments that address the unique mental health needs of a...
-
Clinicians at a nursing home facility want to see if giving residents a plant to tend to will help lower depression. To test this idea, the residents are randomly assigned to one of three groups:...
-
Please give a real-life example of a civil claim(Pleading) that contains the proper name(Legal Name) of the two parties and the amount of the claim.
-
An elderly rancher died and left her estate to her three children. She bequeathed her 17 prize horses in the following manner: \(1 / 2\) to the eldest, \(1 / 3\) to the second child, and \(1 / 9\) to...
-
Twelve people serve on a board and are considering three alternatives: A, B, and C. Here are the choices followed by vote: Determine the winner, if any, using the voting methods in Problems 39-44....
-
A group of fun-loving people have decided to play a practical joke on one of their friends, but they can't decide which friend, Alice (A), Betty (B), or Connie (C). Their preferences are: Use this...
-
Make up an example of a vote that is not transitive.
-
The township of Bella Rosa is divided into two districts, uptown (pop. 16,980) and downtown (pop. 3,350) and is governed by 100 council members. Use this information in Problems 48-53. What is the...
-
Oppositely charged ions attract and this forms salt crystals. What do you think has happened to the salt after mixing with water and how are the water molecules involved? Draw a cartoon picture of...
-
Fahrad Inc. sells all of its product on account. Fahrad has the following accounts receivable payment experience: Percent paid in the month of sale .........10 Percent paid in the month after the...
-
The nominal yield on 6-month T-bills is 7%, while default-free Japanese bonds that mature in 6 months have a nominal rate of 5.5%. In the spot exchange market, 1 yen equals $0.009. If interest rate...
-
Kasperov Corporation has an unlevered cost of equity of 12% and is taxed at a 40% rate. The 4-year forecasts of free cash flow and interest expenses are shown below. Free cash flow and interest...
-
In what circumstances is the compressed adjusted present value (APV) model useful and how would it be applied?
-
The properties of water and steam become identical at (a) 0.1 bar (b) 1 bar (c) \(100 \mathrm{bar}\) (d) 212.2 bar.
-
Steam from an engine exhaust at 1.25 bar flows steadily through an electric calorimeter and comes out at \(1 \mathrm{bar}, 130^{\circ} \mathrm{C}\). The calorimeter has two \(\mathrm{kW}\) heaters...
-
In thermodynamic analysis, a pure substance is that which (a) Consists of only a single chemical species (b) has no dissolved impurities (c) may have a number of chemical species but the composition...

Study smarter with the SolutionInn App


