Oppositely charged ions attract and this forms salt crystals. What do you think has happened to the
Question:
Oppositely charged ions attract and this forms salt crystals. What do you think has happened to the salt after mixing with water and how are the water molecules involved?
Draw a cartoon picture of what you imagine the salt (NaCl) might look like before it is mixed with water and then after mixing with water. To insert your drawing:
Hand-draw on paper
Q2. How would you describe the (observed or imagined) behavior of oil in water when the two liquids are mixed together?
Q3. Interactions between molecules are determined by their intermolecular forces. The molecule below is similar to those found in cooking oil. Looking at this structure, can you make any suggestions for why oil and water do not mix?
Worksheet for Solubility and Molecular Structure
1) Add the solubility data for your lab group to Table 1. Indicate the result for each mixing with "S" for soluble and "I" for insoluble.
Table 1. Group Solubility Data
| Solvents | |||
| Solids (Solutes) |
Water |
Ethyl alcohol |
Petroleum ether |
Biphenyl | I | S | S |
Benzoic acid | I | S | I |
Resorcinol | S | S | I |
Sodium benzoate | S | S | I |
Sodium dihydrogen phosphate | S | I | I |
4. Explain in words how molecular structure relates to solubility. Refer to specific examples in Table 1 to support your argument and show how you reached your conclusions. You should bring in the polarity of the solvent and solid molecules, as well as the specific types of intermolecular forces involved, into your explanation. ?
5) Applying the concepts learned: Which solute should dissolve in which solvent?
On Table 2 predict which solvent will dissolve each solute by typing the solvent letter in the solute box on the table. Briefly give the reasons for your choice of pairings (below the table).
Table 2. Solubility predictions
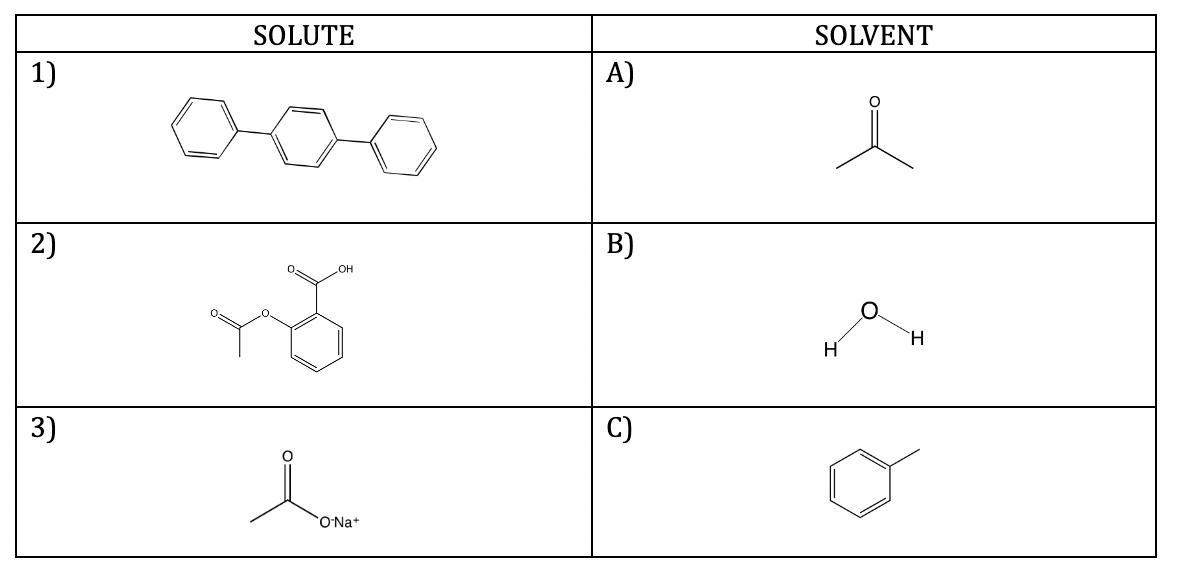
6. Draw the structures of two molecules from Table 1 or Table 2 that can interact through hydrogen bonding. Show partial charges on the appropriate atoms and a dashed line to indicate the interaction. Insert the finished drawing below.

Financial Reporting and Analysis
ISBN: 978-1259722653
7th edition
Authors: Lawrence Revsine, Daniel Collins, Bruce Johnson, Fred Mittelstaedt, Leonard Soffer




